Luminal ATP stimulates fluid and HCO3- secretion in guinea-pig pancreatic duct
- PMID: 10457070
- PMCID: PMC2269526
- DOI: 10.1111/j.1469-7793.1999.0551m.x
Luminal ATP stimulates fluid and HCO3- secretion in guinea-pig pancreatic duct
Abstract
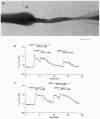
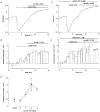
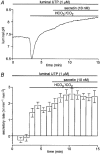
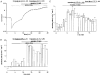
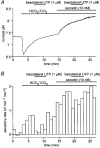
1. The location of purinoceptors in the pancreatic duct and their role in regulating ductal secretion have been investigated by applying ATP and UTP to basolateral and luminal surfaces of pancreatic ducts isolated from the guinea-pig pancreas. 2. Changes in intracellular Ca2+ concentration were measured by microfluorometry in microperfused interlobular duct segments. Fluid and HCO3- secretion were estimated by monitoring luminal pH and luminal volume in sealed duct segments microinjected with BCECF-dextran. 3. Both ATP and UTP (1 microM) caused biphasic increases in intracellular Ca2+ concentration in pancreatic duct cells when applied to either the basolateral or luminal membrane. 4. Luminal application of both ATP and UTP evoked fluid and HCO3- secretion. The maximum response to 1 microM ATP or UTP was about 75 % of that evoked by secretin. By contrast, basolateral application of ATP or UTP inhibited spontaneous secretion by 52 % and 73 %, respectively, and secretin-evoked secretion by 41 % and 38 %, respectively. 5. The data suggest that luminal nucleotides may act in an autocrine or paracrine fashion to enhance ductal secretion while basolateral nucleotides, perhaps released from nerve terminals, may have an inhibitory effect. The fact that both apical and basolateral purinoceptors elevate intracellular Ca2+, but that they have opposite effects on secretion, suggests that additional signalling pathways are involved.
Figures
Similar articles
-
Physiology and pathophysiology of bicarbonate secretion by pancreatic duct epithelium.Nagoya J Med Sci. 2012 Feb;74(1-2):1-18. Nagoya J Med Sci. 2012. PMID: 22515107 Free PMC article. Review.
-
Bicarbonate and fluid secretion evoked by cholecystokinin, bombesin and acetylcholine in isolated guinea-pig pancreatic ducts.J Physiol. 2001 Sep 15;535(Pt 3):795-807. doi: 10.1111/j.1469-7793.2001.00795.x. J Physiol. 2001. PMID: 11559776 Free PMC article.
-
Fluid secretion in interlobular ducts isolated from guinea-pig pancreas.J Physiol. 1998 Sep 1;511 ( Pt 2)(Pt 2):407-22. doi: 10.1111/j.1469-7793.1998.407bh.x. J Physiol. 1998. PMID: 9706019 Free PMC article.
-
CO2 permeability and bicarbonate transport in microperfused interlobular ducts isolated from guinea-pig pancreas.J Physiol. 2000 Oct 15;528 Pt 2(Pt 2):305-15. doi: 10.1111/j.1469-7793.2000.00305.x. J Physiol. 2000. PMID: 11034620 Free PMC article.
-
Mechanisms of bicarbonate secretion in the pancreatic duct.Annu Rev Physiol. 2005;67:377-409. doi: 10.1146/annurev.physiol.67.031103.153247. Annu Rev Physiol. 2005. PMID: 15709963 Review.
Cited by
-
The cystic fibrosis of exocrine pancreas.Cold Spring Harb Perspect Med. 2013 May 1;3(5):a009746. doi: 10.1101/cshperspect.a009746. Cold Spring Harb Perspect Med. 2013. PMID: 23637307 Free PMC article. Review.
-
Trypsin reduces pancreatic ductal bicarbonate secretion by inhibiting CFTR Cl⁻ channels and luminal anion exchangers.Gastroenterology. 2011 Dec;141(6):2228-2239.e6. doi: 10.1053/j.gastro.2011.08.039. Epub 2011 Sep 3. Gastroenterology. 2011. PMID: 21893120 Free PMC article.
-
Physiology and pathophysiology of bicarbonate secretion by pancreatic duct epithelium.Nagoya J Med Sci. 2012 Feb;74(1-2):1-18. Nagoya J Med Sci. 2012. PMID: 22515107 Free PMC article. Review.
-
Pattern of Ca2+ increase determines the type of secretory mechanism activated in dog pancreatic duct epithelial cells.J Physiol. 2006 Oct 1;576(Pt 1):163-78. doi: 10.1113/jphysiol.2006.114876. Epub 2006 Jul 20. J Physiol. 2006. PMID: 16857709 Free PMC article.
-
Purinergic receptors and gastrointestinal secretomotor function.Purinergic Signal. 2008 Sep;4(3):213-36. doi: 10.1007/s11302-008-9104-4. Epub 2008 Jul 5. Purinergic Signal. 2008. PMID: 18604596 Free PMC article.
References
-
- Case RM, Argent BE. Pancreatic duct cell secretion: control and mechanisms of transport. In: Go VLW, Dimagno EP, Gardner JD, Lebenthal E, Reber HA, Scheele GA, editors. The Pancreas: Biology, Pathophysiology, and Disease. 2. New York: Raven Press; 1993. pp. 301–350.
-
- Chan HC, Cheung WT, Leung PY, Wu LJ, Cheng Chew SB, Ko WH, Wong PYD. Purinergic regulation of anion secretion by cystic fibrosis pancreatic duct cells. American Journal of Physiology. 1996;271:C469–477. - PubMed
-
- Christoffersen BC, Hug MJ, Novak I. Different purinergic receptors lead to intracellular calcium increases in pancreatic ducts. Pflügers Archiv. 1998;436:33–39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

