Development of diagnostic reagents to differentiate between Mycobacterium bovis BCG vaccination and M. bovis infection in cattle
- PMID: 10473516
- PMCID: PMC95753
- DOI: 10.1128/CDLI.6.5.675-682.1999
Development of diagnostic reagents to differentiate between Mycobacterium bovis BCG vaccination and M. bovis infection in cattle
Abstract
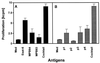
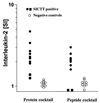
In Great Britain a recent independent scientific review for the government has concluded that the development of a cattle vaccine against Mycobacterium bovis holds the best long-term prospect for tuberculosis control in British herds. A sine qua non for vaccination is the development of a complementary diagnostic test to differentiate between vaccinated animals and those infected with M. bovis so that test-and-slaughter-based control strategies can continue alongside vaccination. In order to assess the feasibility of developing a differential diagnostic test for a live vaccine, we chose M. bovis BCG Pasteur as a model system. Recombinant forms of antigens which are expressed in M. bovis but not, or only at low levels, in BCG Pasteur (ESAT-6, MPB64, MPB70, and MPB83) were produced. These reagents were tested either alone or in combination by using peripheral blood mononuclear cells from M. bovis-infected, BCG-vaccinated, and Mycobacterium avium-sensitized calves. All four antigens induced in vitro proliferation and gamma interferon responses only in M. bovis-infected animals. A cocktail composed of ESAT-6, MPB64, and MPB83 identified infected animals but not those vaccinated with BCG. In addition, promiscuous T-cell epitopes of ESAT-6, MPB64, and MPB83 were formulated into a peptide cocktail. In T-cell assays with this peptide cocktail, infected animals were identified with frequencies similar to those obtained in assays with the protein cocktail, while BCG-vaccinated or M. avium-sensitized animals did not respond. In summary, our results suggest that peptide and protein cocktails can be designed to discriminate between M. bovis infection and BCG vaccination.
Figures
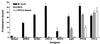
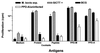
Similar articles
-
Toward the development of diagnostic assays to discriminate between Mycobacterium bovis infection and bacille Calmette-Guérin vaccination in cattle.Clin Infect Dis. 2000 Jun;30 Suppl 3:S291-8. doi: 10.1086/313877. Clin Infect Dis. 2000. PMID: 10875803
-
Use of synthetic peptides derived from the antigens ESAT-6 and CFP-10 for differential diagnosis of bovine tuberculosis in cattle.Clin Diagn Lab Immunol. 2001 May;8(3):571-8. doi: 10.1128/CDLI.8.3.571-578.2001. Clin Diagn Lab Immunol. 2001. PMID: 11329460 Free PMC article.
-
Differentiation between Mycobacterium bovis BCG-vaccinated and M. bovis-infected cattle by using recombinant mycobacterial antigens.Clin Diagn Lab Immunol. 1999 Jan;6(1):1-5. doi: 10.1128/CDLI.6.1.1-5.1999. Clin Diagn Lab Immunol. 1999. PMID: 9874655 Free PMC article.
-
Towards more accurate diagnosis of bovine tuberculosis using defined antigens.Tuberculosis (Edinb). 2001;81(1-2):65-9. doi: 10.1054/tube.2000.0273. Tuberculosis (Edinb). 2001. PMID: 11463225 Review.
-
DIVA reagents for bovine tuberculosis vaccines in cattle.Expert Rev Vaccines. 2011 Jul;10(7):1083-91. doi: 10.1586/erv.11.22. Expert Rev Vaccines. 2011. PMID: 21806401 Review.
Cited by
-
Defined Antigen Skin Test for Bovine Tuberculosis Retains Specificity on Revaccination With Bacillus Calmette-Guérin.Front Vet Sci. 2022 Apr 13;9:814227. doi: 10.3389/fvets.2022.814227. eCollection 2022. Front Vet Sci. 2022. PMID: 35498753 Free PMC article.
-
Recognition of mycobacterial antigens delivered by genetically detoxified Bordetella pertussis adenylate cyclase by T cells from cattle with bovine tuberculosis.Infect Immun. 2004 Nov;72(11):6255-61. doi: 10.1128/IAI.72.11.6255-6261.2004. Infect Immun. 2004. PMID: 15501751 Free PMC article.
-
Interpretation of the gamma interferon test for diagnosis of subclinical paratuberculosis in cattle.Clin Diagn Lab Immunol. 2002 Mar;9(2):453-60. doi: 10.1128/cdli.9.2.453-460.2002. Clin Diagn Lab Immunol. 2002. PMID: 11874893 Free PMC article.
-
What animal models teach humans about tuberculosis.Am J Respir Cell Mol Biol. 2008 Nov;39(5):503-8. doi: 10.1165/rcmb.2008-0154TR. Epub 2008 Jun 12. Am J Respir Cell Mol Biol. 2008. PMID: 18556589 Free PMC article. Review.
-
Evaluation of the Efficacy of BCG in Protecting Against Contact Challenge With Bovine Tuberculosis in Holstein-Friesian and Zebu Crossbred Calves in Ethiopia.Front Vet Sci. 2021 Jul 22;8:702402. doi: 10.3389/fvets.2021.702402. eCollection 2021. Front Vet Sci. 2021. PMID: 34368285 Free PMC article.
References
-
- Anonymous. New Zealand: Bovigam™, it is official. Vet Diagn Newsl. 1997;2:1–3.
-
- Berggren S A. Field experiment with BCG vaccine in Malawi. Br Vet J. 1981;137:88–94. - PubMed
-
- Buddle B M, de Lisle G W, Pfeffer A, Aldwell F E. Immunological responses and protection against Mycobacterium bovis in calves vaccinated with a low dose of BCG. Vaccine. 1995;13:1123–1130. - PubMed
-
- Buddle B M, Keen D, Thomson A, Jowett G, McCarthy A R, Heslop J, De Lisle G W, Stanford J L, Aldwell F E. Protection of cattle from bovine tuberculosis by vaccination with BCG by the respiratory or subcutaneous route, but not by vaccination with killed Mycobacterium vaccae. Res Vet Sci. 1995;59:10–16. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

