Role of alpha(v) integrins in adenovirus cell entry and gene delivery
- PMID: 10477314
- PMCID: PMC103752
- DOI: 10.1128/MMBR.63.3.725-734.1999
Role of alpha(v) integrins in adenovirus cell entry and gene delivery
Abstract
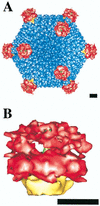
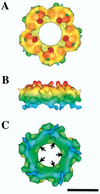
Adenoviruses (Ad) are a significant cause of acute infections in humans; however, replication-defective forms of this virus are currently under investigation for human gene therapy. Approximately 20 to 25% of all the gene therapy trials (phases I to III) conducted over the past 10 years involve the use of Ad gene delivery for treatment inherited or acquired diseases. At present, the most promising applications involve the use of Ad vectors to irradicate certain nonmetastatic tumors and to promote angiogenesis in order to alleviate cardiovascular disease. While specific problems of using Ad vectors remain to be overcome (as is true for almost all viral and nonviral delivery methods), a distinct advantage of Ad is the extensive knowledge of its macromolecular structure, genome organization, sequence, and mode of replication. Moreover, significant information has also been acquired on the interaction of Ad particles with distinct host cell receptors, events which strongly affect virus tropism. This review provides an overview of the structure and function of Ad attachment (coxsackievirus and Ad receptor [CAR]) and internalization (alpha(v) integrins) receptors and discusses their precise role in virus infection and gene delivery. Recent structure studies of integrin-Ad complexes by cryoelectron microscopy are also highlighted. Finally, unanswered questions arising from the current state of knowledge of Ad-receptor interactions are presented in the context of improving Ad vectors for future human gene therapy applications.
Figures





References
-
- Acharya R, Fry E, Stuart D, Fox G, Rowlands D, Brown F. The three-dimensional structure of foot-and-mouth disease virus at 2.9 Å resolution. Nature. 1989;337:709–716. - PubMed
-
- Akke M, Liu J, Cavanagh J, Erickson H P, Palmer A G., III Pervasive conformational fluctuations on microsecond time scales in a fibronectin type III domain. Nat Struct Biol. 1998;5:55–59. - PubMed
-
- Bergelson J M, Cunningham J A, Droguett G, Kurt-Jones E A, Krithivas A, Hong J S, Horwitz M S, Crowell R L, Finberg R W. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science. 1997;275:1320–1323. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

