Vaccinia virus envelope D8L protein binds to cell surface chondroitin sulfate and mediates the adsorption of intracellular mature virions to cells
- PMID: 10482629
- PMCID: PMC112896
- DOI: 10.1128/JVI.73.10.8750-8761.1999
Vaccinia virus envelope D8L protein binds to cell surface chondroitin sulfate and mediates the adsorption of intracellular mature virions to cells
Abstract
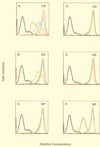
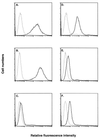
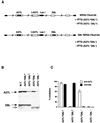
We previously showed that an envelope A27L protein of intracellular mature virions (IMV) of vaccinia virus binds to cell surface heparan sulfate during virus infection. In the present study we identified another viral envelope protein, D8L, that binds to chondroitin sulfate on cells. Soluble D8L protein interferes with the adsorption of wild-type vaccinia virions to cells, indicating a role in virus entry. To explore the interaction of cell surface glycosaminoglycans and vaccinia virus, we generated mutant viruses from a control virus, WR32-7/Ind14K (A27L(+) D8L(+)) to be defective in expression of either the A27L or the D8L gene (A27L(+) D8L(-) or A27L(-) D8L(+)) or both (A27L(-) D8L(-)). The A27L(+) D8L(+) and A27L(-) D8L(+) mutants grew well in BSC40 cells, consistent with previous observations. However, the IMV titers of A27L(+) D8L(-) and A27L(-) D8L(-) viruses in BSC40 cells were reduced, reaching only 10% of the level for the control virus. The data suggested an important role for D8L protein in WR32-7/Ind14K virus growth in cell cultures. A27L protein, on the other hand, could not complement the functions of D8L protein. The low titers of the A27L(+) D8L(-) and A27L(-) D8L(-) mutant viruses were not due to defects in the morphogenesis of IMV, and the mutant virions demonstrated a brick shape similar to that of the control virions. Furthermore, the infectivities of the A27L(+) D8L(-) and A27L(-) D8L(-) mutant virions were 6 to 10% of that of the A27L(+) D8L(+) control virus. Virion binding assays revealed that A27L(+) D8L(-) and A27L(-) D8L(-) mutant virions bound less well to BSC40 cells, indicating that binding of viral D8L protein to cell surface chondroitin sulfate could be important for vaccinia virus entry.
Figures




References
-
- Appleyard G, Hapel A J, Boulter E A. An antigenic difference between intracellular and extracellular rabbitpox virus. J Gen Virol. 1971;13:9–17. - PubMed
-
- Banfield B W, Leduc Y, Esford L, Visalli R J, Brandt C R, Tufaro F. Evidence for an interaction of herpes simplex virus with chondroitin sulfate proteoglycans during infection. Virology. 1995;208:531–539. - PubMed
-
- Byrne K M, Horohov D W, Kousoulas K G. Glycoprotein B of bovine herpesvirus-1 binds heparin. Virology. 1995;209:230–235. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

