Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer's disease
- PMID: 10487842
- PMCID: PMC1866907
- DOI: 10.1016/s0002-9440(10)65184-x
Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer's disease
Abstract
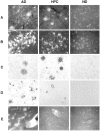
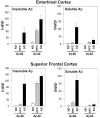
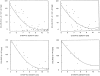
We have characterized amyloid beta peptide (Abeta) concentration, Abeta deposition, paired helical filament formation, cerebrovascular amyloid angiopathy, apolipoprotein E (ApoE) allotype, and synaptophysin concentration in entorhinal cortex and superior frontal gyrus of normal elderly control (ND) patients, Alzheimer's disease (AD) patients, and high pathology control (HPC) patients who meet pathological criteria for AD but show no synapse loss or overt antemortem symptoms of dementia. The measures of Abeta deposition, Abeta-immunoreactive plaques with and without cores, thioflavin histofluorescent plaques, and concentrations of insoluble Abeta, failed to distinguish HPC from AD patients and were poor correlates of synaptic change. By contrast, concentrations of soluble Abeta clearly distinguished HPC from AD patients and were a strong inverse correlate of synapse loss. Further investigation revealed that Abeta40, whether in soluble or insoluble form, was a particularly useful measure for classifying ND, HPC, and AD patients compared with Abeta42. Abeta40 is known to be elevated in cerebrovascular amyloid deposits, and Abeta40 (but not Abeta42) levels, cerebrovascular amyloid angiopathy, and ApoE4 allele frequency were all highly correlated with each other. Although paired helical filaments in the form of neurofibrillary tangles or a penumbra of neurites surrounding amyloid cores also distinguished HPC from AD patients, they were less robust predictors of synapse change compared with soluble Abeta, particularly soluble Abeta40. Previous experiments attempting to relate Abeta deposition to the neurodegeneration that underlies AD dementia may have failed because they assayed the classical, visible forms of the molecule, insoluble neuropil plaques, rather than the soluble, unseen forms of the molecule.
Figures

References
-
- Crystal H, Dickson D, Fuld P, Masur D, Scott R, Mehler M, Masdeu J, Kawas C, Aronson M, Wolfson L: Clinico-pathologic studies in dementia: nondemented subjects with pathologically confirmed Alzheimer’s disease. Neurology 1998, 38:1682-1687 - PubMed
-
- Katzman R, Terry R, DeTeresa R, Brown T, Davis P, Fuld P, Renbing X, Peck A: Clinical, pathological, and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Ann Neurol 1988, 23:138-144 - PubMed
-
- Dickson DW, Crystal HA, Mattiace LA, Masur DM, Blau AD, Davies P, Yen SH, Aronson MK: Identification of normal and pathological aging in prospectively studied nondemented elderly humans. Neurobiol Aging 1991, 13:179-189 - PubMed
-
- Arrigada PA, Marzloff K, Hyman BT: Distribution of Alzheimer-type pathologic changes in nondemented elderly individuals matches the pattern in Alzheimer’s disease. Neurology 1992, 42:1681-1688 - PubMed
-
- Lue LF, Brachova L, Civin WH, Rogers J: Inflammation, Aβ deposition, and neurofibrillary tangle formation as correlates of Alzheimer’s disease neurodegeneration. J Neuropathol Exp Neurol 1996, 55:1083-1088 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

