Characterization and dynamics of aggresome formation by a cytosolic GFP-chimera
- PMID: 10491388
- PMCID: PMC2156127
- DOI: 10.1083/jcb.146.6.1239
Characterization and dynamics of aggresome formation by a cytosolic GFP-chimera
Abstract
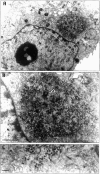
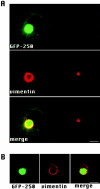
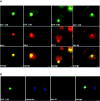
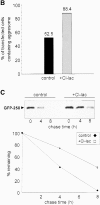
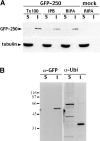
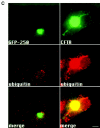
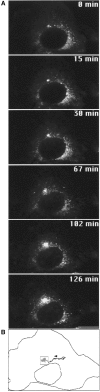
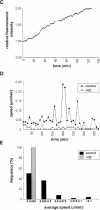
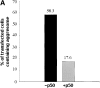
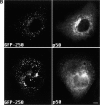
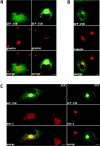
Formation of a novel structure, the aggresome, has been proposed to represent a general cellular response to the presence of misfolded proteins (Johnston, J.A., C.L. Ward, and R.R. Kopito. 1998. J. Cell Biol. 143:1883-1898; Wigley, W.C., R.P. Fabunmi, M.G. Lee, C.R. Marino, S. Muallem, G.N. DeMartino, and P.J. Thomas. 1999. J. Cell Biol. 145:481-490). To test the generality of this finding and characterize aspects of aggresome composition and its formation, we investigated the effects of overexpressing a cytosolic protein chimera (GFP-250) in cells. Overexpression of GFP-250 caused formation of aggresomes and was paralleled by the redistribution of the intermediate filament protein vimentin as well as by the recruitment of the proteasome, and the Hsp70 and the chaperonin systems of chaperones. Interestingly, GFP-250 within the aggresome appeared not to be ubiquitinated. In vivo time-lapse analysis of aggresome dynamics showed that small aggregates form within the periphery of the cell and travel on microtubules to the MTOC region where they remain as distinct but closely apposed particulate structures. Overexpression of p50/dynamitin, which causes the dissociation of the dynactin complex, significantly inhibited the formation of aggresomes, suggesting that the minus-end-directed motor activities of cytoplasmic dynein are required for aggresome formation. Perinuclear aggresomes interfered with correct Golgi localization and disrupted the normal astral distribution of microtubules. However, ER-to-Golgi protein transport occurred normally in aggresome containing cells. Our results suggest that aggresomes can be formed by soluble, nonubiquitinated proteins as well as by integral transmembrane ubiquitinated ones, supporting the hypothesis that aggresome formation might be a general cellular response to the presence of misfolded proteins.
Figures



References
-
- Bercovich Z., Rosenberg-Hasson Y., Ciechanover A., Kahana C. Degradation of ornithine decarboxylase in reticulocyte lysate is ATP-dependent but ubiquitin-independent. J. Biol. Chem. 1989;264:15949–15952. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

