Regulation of melanosome movement in the cell cycle by reversible association with myosin V
- PMID: 10491390
- PMCID: PMC2156116
- DOI: 10.1083/jcb.146.6.1265
Regulation of melanosome movement in the cell cycle by reversible association with myosin V
Abstract
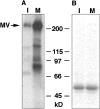
Previously, we have shown that melanosomes of Xenopus laevis melanophores are transported along both microtubules and actin filaments in a coordinated manner, and that myosin V is bound to purified melanosomes (Rogers, S., and V.I. Gelfand. 1998. Curr. Biol. 8:161-164). In the present study, we have demonstrated that myosin V is the actin-based motor responsible for melanosome transport. To examine whether myosin V was regulated in a cell cycle-dependent manner, purified melanosomes were treated with interphase- or metaphase-arrested Xenopus egg extracts and assayed for in vitro motility along Nitella actin filaments. Motility of organelles treated with mitotic extract was found to decrease dramatically, as compared with untreated or interphase extract-treated melanosomes. This mitotic inhibition of motility correlated with the dissociation of myosin V from melanosomes, but the activity of soluble motor remained unaffected. Furthermore, we find that myosin V heavy chain is highly phosphorylated in metaphase extracts versus interphase extracts. We conclude that organelle transport by myosin V is controlled by a cell cycle-regulated association of this motor to organelles, and that this binding is likely regulated by phosphorylation of myosin V during mitosis.
Figures





Similar articles
-
Cell cycle regulation of myosin-V by calcium/calmodulin-dependent protein kinase II.Science. 2001 Aug 17;293(5533):1317-20. doi: 10.1126/science.1061086. Science. 2001. PMID: 11509731
-
Cell biology. A switch to release the motor.Science. 2001 Aug 17;293(5533):1263-4. doi: 10.1126/science.1064602. Science. 2001. PMID: 11509712 No abstract available.
-
Myosin Va's adaptor protein melanophilin enforces track selection on the microtubule and actin networks in vitro.Proc Natl Acad Sci U S A. 2017 Jun 13;114(24):E4714-E4723. doi: 10.1073/pnas.1619473114. Epub 2017 May 30. Proc Natl Acad Sci U S A. 2017. PMID: 28559319 Free PMC article.
-
Class V myosins.Biochim Biophys Acta. 2000 Mar 17;1496(1):36-51. doi: 10.1016/s0167-4889(00)00007-0. Biochim Biophys Acta. 2000. PMID: 10722875 Review. No abstract available.
-
Vesicle transport: the role of actin filaments and myosin motors.Microsc Res Tech. 1999 Oct 15;47(2):93-106. doi: 10.1002/(SICI)1097-0029(19991015)47:2<93::AID-JEMT2>3.0.CO;2-P. Microsc Res Tech. 1999. PMID: 10523788 Review.
Cited by
-
Intermolecular autophosphorylation regulates myosin IIIa activity and localization in parallel actin bundles.J Biol Chem. 2010 Nov 12;285(46):35770-82. doi: 10.1074/jbc.M110.144360. Epub 2010 Sep 7. J Biol Chem. 2010. PMID: 20826793 Free PMC article.
-
Collaboration between Distinct Rab Small GTPase Trafficking Circuits Mediates Bacterial Clearance from the Bladder Epithelium.Cell Host Microbe. 2017 Sep 13;22(3):330-342.e4. doi: 10.1016/j.chom.2017.08.002. Cell Host Microbe. 2017. PMID: 28910634 Free PMC article.
-
Melanosomes transported by myosin-V in Xenopus melanophores perform slow 35 nm steps.Biophys J. 2006 Jan 1;90(1):L07-9. doi: 10.1529/biophysj.105.075556. Epub 2005 Nov 11. Biophys J. 2006. PMID: 16284273 Free PMC article.
-
Mechanical properties of organelles driven by microtubule-dependent molecular motors in living cells.PLoS One. 2011 Apr 1;6(4):e18332. doi: 10.1371/journal.pone.0018332. PLoS One. 2011. PMID: 21483765 Free PMC article.
-
TLRR (lrrc67) interacts with PP1 and is associated with a cytoskeletal complex in the testis.Biol Cell. 2010 Jan 13;102(3):173-89. doi: 10.1042/BC20090091. Biol Cell. 2010. PMID: 19886865 Free PMC article.
References
-
- Adams R., Pollard T. Propulsion of organelles isolated from Acanthamoeba along actin filaments by myosin I. Nature. 1986;322:754–756. - PubMed
-
- Allan V. Assay of membrane motility in interphase and metaphase Xenopus extracts. Methods Cell Biol. 1993;39:203–226. - PubMed
-
- Amaratunga A., Morin P., Kosik K., Fine R. Inhibition of kinesin synthesis and rapid anterograde axonal transport in vivo by an antisense oligonucleotide. J. Biol. Chem. 1993;268:17427–17430. - PubMed

