TGF-beta1 in Aplysia: role in long-term changes in the excitability of sensory neurons and distribution of TbetaR-II-like immunoreactivity
- PMID: 10492013
- PMCID: PMC311291
TGF-beta1 in Aplysia: role in long-term changes in the excitability of sensory neurons and distribution of TbetaR-II-like immunoreactivity
Abstract
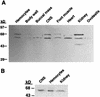
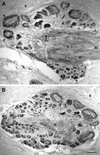
Exogenous recombinant human transforming growth factor beta-1 (TGF-beta1) induced long-term facilitation of Aplysia sensory-motor synapses. In addition, 5-HT-induced facilitation was blocked by application of a soluble fragment of the extracellular portion of the TGF-beta1 type II receptor (TbetaR-II), which presumably acted by scavenging an endogenous TGF-beta1-like molecule. Because TbetaR-II is essential for transmembrane signaling by TGF-beta, we sought to determine whether Aplysia tissues contained TbetaR-II and specifically, whether neurons expressed the receptor. Western blot analysis of Aplysia tissue extracts demonstrated the presence of a TbetaR-II-immunoreactive protein in several tissue types. The expression and distribution of TbetaR-II-immunoreactive proteins in the central nervous system was examined by immunohistochemistry to elucidate sites that may be responsive to TGF-beta1 and thus may play a role in synaptic plasticity. Sensory neurons in the ventral-caudal cluster of the pleural ganglion were immunoreactive for TbetaR-II, as well as many neurons in the pedal, abdominal, buccal, and cerebral ganglia. Sensory neurons cultured in isolation and cocultured sensory and motor neurons were also immunoreactive. TGF-beta1 affected the biophysical properties of cultured sensory neurons, inducing an increase of excitability that persisted for at least 48 hr. Furthermore, exposure to TGF-beta1 resulted in a reduction in the firing threshold of sensory neurons. These results provide further support for the hypothesis that TGF-beta1 plays a role in long-term synaptic plasticity in Aplysia.
Figures



Similar articles
-
Transforming growth factor beta1 alters synapsin distribution and modulates synaptic depression in Aplysia.J Neurosci. 2002 May 1;22(9):RC220. doi: 10.1523/JNEUROSCI.22-09-j0004.2002. Epub 2002 Apr 24. J Neurosci. 2002. PMID: 11978861 Free PMC article.
-
Role of transforming growth factor-beta in long-term synaptic facilitation in Aplysia.Science. 1997 Feb 28;275(5304):1318-20. doi: 10.1126/science.275.5304.1318. Science. 1997. PMID: 9036859
-
TGF-beta1-induced long-term changes in neuronal excitability in aplysia sensory neurons depend on MAPK.J Neurophysiol. 2006 May;95(5):3286-90. doi: 10.1152/jn.00770.2005. J Neurophysiol. 2006. PMID: 16617179
-
The spatiotemporal expression of TGF-beta1 and its receptors during periosteal chondrogenesis in vitro.J Orthop Res. 2002 May;20(3):562-74. doi: 10.1016/S0736-0266(01)00130-9. J Orthop Res. 2002. PMID: 12038632
-
Postsynaptic regulation of the development and long-term plasticity of Aplysia sensorimotor synapses in cell culture.J Neurobiol. 1994 Jun;25(6):666-93. doi: 10.1002/neu.480250608. J Neurobiol. 1994. PMID: 8071666 Review.
Cited by
-
Coregulation of glutamate uptake and long-term sensitization in Aplysia.J Neurosci. 2004 Oct 6;24(40):8829-37. doi: 10.1523/JNEUROSCI.2167-04.2004. J Neurosci. 2004. PMID: 15470149 Free PMC article.
-
The membrane trafficking and functionality of the K+-Cl- co-transporter KCC2 is regulated by TGF-β2.J Cell Sci. 2016 Sep 15;129(18):3485-98. doi: 10.1242/jcs.189860. Epub 2016 Aug 5. J Cell Sci. 2016. PMID: 27505893 Free PMC article.
-
Serotonin stimulates phosphorylation of Aplysia synapsin and alters its subcellular distribution in sensory neurons.J Neurosci. 2002 Jul 1;22(13):5412-22. doi: 10.1523/JNEUROSCI.22-13-05412.2002. J Neurosci. 2002. PMID: 12097493 Free PMC article.
-
Role of p90 ribosomal S6 kinase in long-term synaptic facilitation and enhanced neuronal excitability.Sci Rep. 2020 Jan 17;10(1):608. doi: 10.1038/s41598-020-57484-y. Sci Rep. 2020. PMID: 31953461 Free PMC article.
-
Transforming growth factor beta induces sensory neuronal hyperexcitability, and contributes to pancreatic pain and hyperalgesia in rats with chronic pancreatitis.Mol Pain. 2012 Sep 11;8:65. doi: 10.1186/1744-8069-8-65. Mol Pain. 2012. PMID: 22963239 Free PMC article.
References
-
- Ata AK, Funa K, Olsson Y. Expression of various TGF-β isoforms and type I receptor in necrotizing human brain lesions. Acta Neuropathol. 1997;93:326–333. - PubMed
-
- Bailey CH, Montarolo PG, Chen M, Kandel ER, Schacher S. Inhibitors of protein and RNA synthesis block structural changes that accompany long-term synaptic plasticity in Aplysia. Neuron. 1992;9:749–758. - PubMed
-
- Barnard JA, Lyons RM, Moses HL. The cell biology of transforming growth factor β. Biochim Biophys Acta. 1990;1032:79–87. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
