Use of in vivo-regulated promoters to deliver antigens from attenuated Salmonella enterica var. Typhimurium
- PMID: 10496887
- PMCID: PMC96862
- DOI: 10.1128/IAI.67.10.5133-5141.1999
Use of in vivo-regulated promoters to deliver antigens from attenuated Salmonella enterica var. Typhimurium
Abstract
This study describes the construction and analysis of three in vivo-inducible promoter expression plasmids, containing pnirB, ppagC, and pkatG, for the delivery of foreign antigens in the DeltaaroAD mutant of Salmonella enterica var. Typhimurium (hereafter referred to as S. typhimurium). The reporter genes encoding beta-galactosidase and firefly luciferase were used to assess the comparative levels of promoter activity in S. typhimurium in vitro in response to different induction stimuli and in vivo in immunized mice. It was determined that the ppagC construct directed the expression of more beta-galactosidase and luciferase in S. typhimurium than the pnirB and pkatG constructs, both in vitro and in vivo. The gene encoding the C fragment of tetanus toxin was expressed in the aroAD mutant of S. typhimurium (BRD509) under the control of the three promoters. Mice orally immunized with attenuated S. typhimurium expressing C fragment under control of the pagC promoter [BRD509(pKK/ppagC/C frag)] mounted the highest tetanus toxoid-specific serum antibody response. Levels of luciferase expression in vivo and C-fragment expression in vitro from the pagC promoter appeared to be equivalent to if not lower than the levels of expression detected with the constitutive trc promoter. However, mice immunized with BRD509(pKK/ppagC/C frag) induced significantly higher levels of tetanus toxoid-specific antibody than BRD509(pKK/C frag)-immunized mice, suggesting that the specific location of foreign antigen expression may be important for immunogenicity. Mutagenesis of the ribosome binding sites (RBS) in the three promoter/C fragment expression plasmids was also performed. Despite optimization of the RBS in the three different promoter elements, the expression levels in vivo and overall immunogenicity of C fragment when delivered to mice by attenuated S. typhimurium were not affected. These studies suggest that in vivo-inducible promoters may give rise to enhanced immunogenicity and increase the efficacy of S. typhimurium as a vaccine vector.
Figures

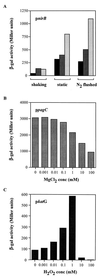
 ),
nitrite (2.5 mM) (░⃞), or no supplement (■) was added to the growth
media. (B) BRD509(pKK/ppagC/lacZ) cultures were induced for
2.5 h by the addition of 0.001 to 100 mM MgCl2. (C)
BRD509(pKK/pkatG/lacZ) cultures were induced for 2.5 h
by the addition of 0.001 to 100 mM H2O2.
β-Galactosidase activity is expressed in Miller units. The results
presented are representative of a number of separate experiments.
),
nitrite (2.5 mM) (░⃞), or no supplement (■) was added to the growth
media. (B) BRD509(pKK/ppagC/lacZ) cultures were induced for
2.5 h by the addition of 0.001 to 100 mM MgCl2. (C)
BRD509(pKK/pkatG/lacZ) cultures were induced for 2.5 h
by the addition of 0.001 to 100 mM H2O2.
β-Galactosidase activity is expressed in Miller units. The results
presented are representative of a number of separate experiments.



Similar articles
-
Oral vaccination against tetanus: comparison of the immunogenicities of Salmonella strains expressing fragment C from the nirB and htrA promoters.Infect Immun. 1998 Jul;66(7):3080-7. doi: 10.1128/IAI.66.7.3080-3087.1998. Infect Immun. 1998. PMID: 9632569 Free PMC article.
-
In vitro and in vivo stability of recombinant plasmids in a vaccine strain of Salmonella enterica var. Typhimurium.FEMS Immunol Med Microbiol. 2003 Jul 15;37(2-3):111-9. doi: 10.1016/S0928-8244(03)00065-8. FEMS Immunol Med Microbiol. 2003. PMID: 12832114
-
Prior immunity to homologous and heterologous Salmonella serotypes suppresses local and systemic anti-fragment C antibody responses and protection from tetanus toxin in mice immunized with Salmonella strains expressing fragment C.Infect Immun. 1999 Aug;67(8):3810-5. doi: 10.1128/IAI.67.8.3810-3815.1999. Infect Immun. 1999. PMID: 10417142 Free PMC article.
-
A murine model of intranasal immunization to assess the immunogenicity of attenuated Salmonella typhi live vector vaccines in stimulating serum antibody responses to expressed foreign antigens.Vaccine. 1997 Apr-May;15(6-7):700-8. doi: 10.1016/s0264-410x(96)00227-7. Vaccine. 1997. PMID: 9178472
-
Induction of protective immunity against Streptococcus mutans colonization after mucosal immunization with attenuated Salmonella enterica serovar typhimurium expressing an S. mutans adhesin under the control of in vivo-inducible nirB promoter.Infect Immun. 2001 Apr;69(4):2154-61. doi: 10.1128/IAI.69.4.2154-2161.2001. Infect Immun. 2001. PMID: 11254570 Free PMC article.
Cited by
-
A sopB deletion mutation enhances the immunogenicity and protective efficacy of a heterologous antigen delivered by live attenuated Salmonella enterica vaccines.Infect Immun. 2008 Nov;76(11):5238-46. doi: 10.1128/IAI.00720-08. Epub 2008 Sep 2. Infect Immun. 2008. PMID: 18765737 Free PMC article.
-
Construction, characterization, and immunogenicity of an attenuated Salmonella enterica serovar typhimurium pgtE vaccine expressing fimbriae with integrated viral epitopes from the spiC promoter.Infect Immun. 2003 Aug;71(8):4664-73. doi: 10.1128/IAI.71.8.4664-4673.2003. Infect Immun. 2003. PMID: 12874347 Free PMC article.
-
Scalable Reporter Assays to Analyze the Regulation of stx2 Expression in Shiga Toxin-Producing Enteropathogens.Toxins (Basel). 2021 Jul 29;13(8):534. doi: 10.3390/toxins13080534. Toxins (Basel). 2021. PMID: 34437405 Free PMC article.
-
Influence of promoter, gene copy number, and preexisting immunity on humoral and cellular responses to a vectored antigen delivered by a Salmonella enterica vaccine.Clin Vaccine Immunol. 2009 Jan;16(1):78-87. doi: 10.1128/CVI.00253-08. Epub 2008 Nov 12. Clin Vaccine Immunol. 2009. PMID: 19005022 Free PMC article.
-
Single dose novel Salmonella vaccine enhances resistance against visceralizing L. major and L. donovani infection in susceptible BALB/c mice.PLoS Negl Trop Dis. 2011 Dec;5(12):e1406. doi: 10.1371/journal.pntd.0001406. Epub 2011 Dec 27. PLoS Negl Trop Dis. 2011. PMID: 22216363 Free PMC article.
References
-
- Anderson R, Dougan G, Roberts M. Delivery of the Pertactin/P.69 polypeptide of Bordetella pertussis using an attenuated Salmonella typhimuriumvaccine strain: expression levels and immune response. Vaccine. 1996;14:1384–1390. - PubMed
-
- Bell A I, Cole J A, Busby S J. Molecular genetic analysis of an FNR-dependent anaerobically inducible Escherichia colipromoter. Mol Microbiol. 1990;4:1753–1763. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

