[Phe1psi(CH2-NH)Gly2]nociceptin-(1 - 13)-NH2 activation of an inward rectifier as a partial agonist of ORL1 receptors in rat periaqueductal gray
- PMID: 10498840
- PMCID: PMC1571592
- DOI: 10.1038/sj.bjp.0702746
[Phe1psi(CH2-NH)Gly2]nociceptin-(1 - 13)-NH2 activation of an inward rectifier as a partial agonist of ORL1 receptors in rat periaqueductal gray
Abstract
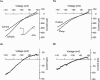
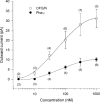
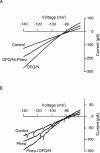
1. [Phe1psi(CH2-NH)Gly2]nociceptin-(1 - 13)-NH2 (Phepsi), a tridecapeptide analogue of orphanin FQ/nociceptin (OFQ/N), was introduced as a competitive antagonist of opioid receptor-like orphan receptor (ORL1) in guinea-pig ileum and mouse vas deferens preparations in vitro but was recently found to act as an agonist in vivo. 2. In the periaqueductal gray, a site enriched with both OFQ/N and ORL1 and involved in OFQ/N-induced hyperalgesia and anti-analgesia, the effects of Phepsi and OFQ/N on the membrane current were studied using whole cell patch clamp recording technique in rat brain slices. 3. OFQ/N (0.01 - 1 microM) activated an inwardly rectifying type of K+ channels in ventrolateral neurons of PAG. Phepsi (0.03 - 1 microM), like OFQ/N, also activated this inward rectifier but had only 30% efficacy of OFQ/N. 4 At maximal effective concentration (1 microM), Phepsi reversed the increment of K+ conductance induced by OFQ/N (300 nM) by 46%. On the other hand, Phepsi also prevented the effect of OFQ/N if pretreated before OFQ/N. 5 It is suggested that Phepsi acts as a partial agonist of ORL1 that mediates the activation of inwardly rectifying K+ channels in ventrolateral neurons of rat periaqueductal gray.
Figures
Similar articles
-
[Nphe(1)]N/OFQ-(1-13)-NH(2) is a competitive and selective antagonist at nociceptin/orphanin FQ receptors mediating K(+) channel activation in rat periaqueductal gray slices.Neuropharmacology. 2002 Feb;42(2):246-52. doi: 10.1016/s0028-3908(01)00159-9. Neuropharmacology. 2002. PMID: 11804621
-
Pharmacological characterization of the nociceptin receptor, ORL1. Insight from the inward rectifier activation in the periaqueductal gray.J Biomed Sci. 2000 May-Jun;7(3):232-40. doi: 10.1007/BF02255471. J Biomed Sci. 2000. PMID: 10810242 Review.
-
Quantitative study of [(pF)Phe4,Arg14,Lys15]nociceptin/orphanin FQ-NH2 (UFP-102) at NOP receptors in rat periaqueductal gray slices.Eur J Pharmacol. 2008 Jan 28;579(1-3):110-5. doi: 10.1016/j.ejphar.2007.10.006. Epub 2007 Oct 11. Eur J Pharmacol. 2008. PMID: 17976580
-
Ro 64-6198 [(1S,3aS)-8-(2,3,3a,4,5,6-Hexahydro-1H-phenalen-1-yl)-1-phenyl-1,3,8-triaza-spiro[4.5]decan-4-one] acts differently from nociceptin/orphanin FQ in rat periaqueductal gray slices.J Pharmacol Exp Ther. 2004 Nov;311(2):645-51. doi: 10.1124/jpet.104.070219. Epub 2004 Jul 13. J Pharmacol Exp Ther. 2004. PMID: 15254141
-
Pharmacological characterization of nociceptin/orphanin FQ receptors, a novel opioid receptor family, in the midbrain periaqueductal gray.Ann N Y Acad Sci. 2004 Oct;1025:398-403. doi: 10.1196/annals.1316.049. Ann N Y Acad Sci. 2004. PMID: 15542742 Review.
Cited by
-
Orphanin FQ/nociceptin and [Phe(1)Psi(CH(2)-NH)Gly(2)] nociceptin(1-13)-NH(2) stimulate gastric motor function in anaesthetized rats.Br J Pharmacol. 2000 Aug;130(7):1639-45. doi: 10.1038/sj.bjp.0703463. Br J Pharmacol. 2000. PMID: 10928969 Free PMC article.
-
Characterization of the ORL(1) receptor on adrenergic nerves in the rat anococcygeus muscle.Br J Pharmacol. 2000 Sep;131(2):349-55. doi: 10.1038/sj.bjp.0703583. Br J Pharmacol. 2000. PMID: 10991930 Free PMC article.
-
Role of nociceptin/orphanin FQ and nociceptin opioid peptide receptor in depression and antidepressant effects of nociceptin opioid peptide receptor antagonists.Korean J Physiol Pharmacol. 2019 Nov;23(6):427-448. doi: 10.4196/kjpp.2019.23.6.427. Epub 2019 Oct 24. Korean J Physiol Pharmacol. 2019. PMID: 31680765 Free PMC article. Review.
-
Actions of nociceptin/orphanin FQ and other prepronociceptin products on rat rostral ventromedial medulla neurons in vitro.J Physiol. 2001 Aug 1;534(Pt 3):849-59. doi: 10.1111/j.1469-7793.2001.00849.x. J Physiol. 2001. PMID: 11483714 Free PMC article.
-
Pharmacology of nociceptin and its receptor: a novel therapeutic target.Br J Pharmacol. 2000 Apr;129(7):1261-83. doi: 10.1038/sj.bjp.0703219. Br J Pharmacol. 2000. PMID: 10742280 Free PMC article. Review.
References
-
- ANTON B., FEIN J., TO T., LI X., SILBERSTEIN L., EVANS C.J. Immunohistochemical localization of ORL-1 in the central nervous system of the rat. J. Comp. Neurol. 1996;368:229–251. - PubMed
-
- BEHBEHANI M.M. Functional characteristics of the midbrain periaqueductal gray. Prog. Neurobiol. 1995;46:575–605. - PubMed
-
- BLANTON M.G., LO TURCO J.J., KRIEGSTEIN A.R. Whole cell recording from neurons in slices of reptilian and mammalian cerebral cortex. J. Neurosci. Meth. 1989;30:203–210. - PubMed
-
- BUTOUR J.L., MOISAND C., MOLLEREAU C., MEUNIER J.C. [Phe1ψ(CH2-NH)Gly2]nociceptin-(1-13)-NH2 is an agonist of the nociceptin (ORL1) receptor. Eur. J. Pharmacol. 1998;349:R5–R6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

