Differential effects of caffeine and perchlorate on excitation-contraction coupling in mammalian skeletal muscle
- PMID: 10517813
- PMCID: PMC2269578
- DOI: 10.1111/j.1469-7793.1999.00217.x
Differential effects of caffeine and perchlorate on excitation-contraction coupling in mammalian skeletal muscle
Abstract
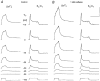
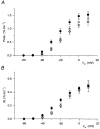
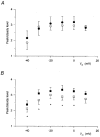
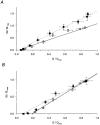
1. Enzymatically dissociated single muscle fibres of the rat were studied under voltage clamp conditions in a double Vaseline gap experimental chamber. Intramembrane charge movement and changes in intracellular calcium concentration ([Ca2+]i) were measured and the rate of calcium release (Rrel) from the sarcoplasmic reticulum (SR) was calculated. This enabled the determination of SR permeability and thus the estimation of the transfer function between intramembrane charge movement and SR permeability. 2. Perchlorate (3 mM) shifted the membrane potential dependence of intramembrane charge movement to more negative voltages without any effect on the steepness or on the maximal available charge. The drug increased SR permeability at every membrane potential but did not alter the peak-to-steady level ratio. It also increased the slope of the transfer function, indicating a more efficient coupling between the voltage sensors and the ryanodine receptors. 3. Caffeine (1 mM), on the other hand, increased SR permeability without altering the voltage dependence of intramembrane charge movement. It neither prolonged the depolarization-induced increase in [Ca2+]i at short pulse durations nor altered the time to peak of Rrel. The augmentation of SR permeability by the drug was more pronounced during the peak caffeine response than during its steady level. This was manifested in a leftward shift of the transfer function rather than an increase in its slope. 4. These observations indicate that perchlorate and caffeine alter the coupling between the voltage sensors and SR calcium release channels in mammalian skeletal muscle. They do not, however, share a common mechanism for enhancing the depolarization-induced release of calcium from the SR.
Figures





Similar articles
-
Effects of tetracaine on sarcoplasmic calcium release in mammalian skeletal muscle fibres.J Physiol. 1999 Mar 15;515 ( Pt 3)(Pt 3):843-57. doi: 10.1111/j.1469-7793.1999.843ab.x. J Physiol. 1999. PMID: 10066909 Free PMC article.
-
Activation of Ca2+ release by caffeine and voltage in frog skeletal muscle.J Physiol. 1996 Jun 1;493 ( Pt 2)(Pt 2):317-39. doi: 10.1113/jphysiol.1996.sp021386. J Physiol. 1996. PMID: 8782099 Free PMC article.
-
Caffeine enhances intramembranous charge movement in frog skeletal muscle by increasing cytoplasmic Ca2+ concentration.J Physiol. 1996 Jun 1;493 ( Pt 2)(Pt 2):341-56. doi: 10.1113/jphysiol.1996.sp021387. J Physiol. 1996. PMID: 8782100 Free PMC article.
-
Intramembrane charge movement and sarcoplasmic calcium release in enzymatically isolated mammalian skeletal muscle fibres.J Physiol. 1997 Dec 1;505 ( Pt 2)(Pt 2):371-84. doi: 10.1111/j.1469-7793.1997.371bb.x. J Physiol. 1997. PMID: 9423180 Free PMC article.
-
Voltage clamp methods for the study of membrane currents and SR Ca(2+) release in adult skeletal muscle fibres.Prog Biophys Mol Biol. 2012 Apr;108(3):98-118. doi: 10.1016/j.pbiomolbio.2012.01.001. Epub 2012 Jan 26. Prog Biophys Mol Biol. 2012. PMID: 22306655 Free PMC article. Review.
Cited by
-
Altered elementary calcium release events and enhanced calcium release by thymol in rat skeletal muscle.Biophys J. 2004 Mar;86(3):1436-53. doi: 10.1016/S0006-3495(04)74213-7. Biophys J. 2004. PMID: 14990472 Free PMC article.
-
Altered inactivation of Ca2+ current and Ca2+ release in mouse muscle fibers deficient in the DHP receptor gamma1 subunit.J Gen Physiol. 2004 Nov;124(5):605-18. doi: 10.1085/jgp.200409168. J Gen Physiol. 2004. PMID: 15504904 Free PMC article.
-
Reciprocal dihydropyridine and ryanodine receptor interactions in skeletal muscle activation.J Muscle Res Cell Motil. 2011 Nov;32(3):171-202. doi: 10.1007/s10974-011-9262-9. Epub 2011 Oct 13. J Muscle Res Cell Motil. 2011. PMID: 21993921 Review.
-
The Penicillium chrysogenum-derived antifungal peptide shows no toxic effects on mammalian cells in the intended therapeutic concentration.Naunyn Schmiedebergs Arch Pharmacol. 2005 Feb;371(2):122-32. doi: 10.1007/s00210-004-1013-7. Epub 2005 Feb 9. Naunyn Schmiedebergs Arch Pharmacol. 2005. PMID: 15702351
-
Deterministic inactivation of calcium release channels in mammalian skeletal muscle.J Physiol. 2000 Nov 1;528(Pt 3):447-56. doi: 10.1111/j.1469-7793.2000.00447.x. J Physiol. 2000. PMID: 11060123 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

