Postsynaptic clustering of gamma-aminobutyric acid type A receptors by the gamma3 subunit in vivo
- PMID: 10536013
- PMCID: PMC23133
- DOI: 10.1073/pnas.96.22.12860
Postsynaptic clustering of gamma-aminobutyric acid type A receptors by the gamma3 subunit in vivo
Abstract
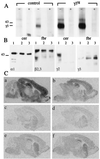
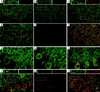
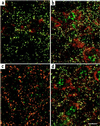
Synaptic localization of gamma-aminobutyric acid type A (GABA(A)) receptors is a prerequisite for synaptic inhibitory function, but the mechanism by which different receptor subtypes are localized to postsynaptic sites is poorly understood. The gamma2 subunit and the postsynaptic clustering protein gephyrin are required for synaptic localization and function of major GABA(A) receptor subtypes. We now show that transgenic overexpression of the gamma3 subunit in gamma2 subunit-deficient mice restores benzodiazepine binding sites, benzodiazepine-modulated whole cell currents, and postsynaptic miniature currents, suggesting the formation of functional, postsynaptic receptors. Moreover, the gamma3 subunit can substitute for gamma2 in the formation of GABA(A) receptors that are synaptically clustered and colocalized with gephyrin in vivo. These clusters were formed even in brain regions devoid of endogenous gamma3 subunit, indicating that the factors present for clustering of gamma2 subunit-containing receptors are sufficient to cluster gamma3 subunit-containing receptors. The GABA(A) receptor and gephyrin-clustering properties of the ectopic gamma3 subunit were also observed for the endogenous gamma3 subunit, but only in the absence of the gamma2 subunit, suggesting that the gamma3 subunit is at a competitive disadvantage with the gamma2 subunit for clustering of postsynaptic GABA(A) receptors in wild-type mice.
Figures

References
-
- Kirsch J, Meyer G, Betz H. Mol Cell Neurosci. 1996;8:93–98. - PubMed
-
- Mohler H, Fritschy J-M, Lüscher B, Rudolph U, Benson J, Benke D. In: Ion Channels. Narahashi T, editor. Vol. 4. New York: Plenum; 1996. pp. 89–113. - PubMed
-
- Sieghart W. Pharmacol Rev. 1995;47:181–234. - PubMed
-
- Macdonald R L, Olsen R W. Annu Rev Neurosci. 1994;17:569–602. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

