Accumulation of 3-ketosteroids induced by itraconazole in azole-resistant clinical Candida albicans isolates
- PMID: 10543744
- PMCID: PMC89540
- DOI: 10.1128/AAC.43.11.2663
Accumulation of 3-ketosteroids induced by itraconazole in azole-resistant clinical Candida albicans isolates
Abstract
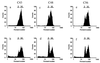
The effects of itraconazole on ergosterol biosynthesis were investigated in a series of 16 matched clinical Candida albicans isolates which had been previously analyzed for mechanisms of resistance to azoles (D. Sanglard, K. Kuchler, F. Ischer, J. L. Pagani, M. Monod, and J. Bille, Antimicrob. Agents Chemother., 39:2378-2386, 1995). Under control conditions, all isolates contained ergosterol as the predominant sterol, except two strains (C48 and C56). In isolates C48 and C56, both less susceptible to azoles than their parent, C43, substantial concentrations (20 to 30%) of 14alpha-methyl-ergosta-8,24(28)-diene-3beta,6alpha-dio l (3, 6-diol) were found. Itraconazole treatment of C43 resulted in a dose-dependent inhibition of ergosterol biosynthesis (50% inhibitory concentration, 2 nM) and accumulation of 3,6-diol (up to 60% of the total sterols) together with eburicol, lanosterol, obtusifoliol, 14alpha-methyl-ergosta-5,7,22,24(28)-tetraene-3betaol, and 14alpha-methyl-fecosterol. In strains C48 and C56, no further increase of 3,6-diol was observed after exposure to itraconazole. Ergosterol synthesis was less sensitive to itraconazole inhibition, as was expected for these azole-resistant isolates which overexpress ATP-binding cassette transporter genes CDR1 and CDR2. In addition to 3,6-diol, substantial amounts of obtusifolione were found after exposure to itraconazole. This toxic 3-ketosteroid was demonstrated previously to accumulate after itraconazole treatment in Cryptococcus neoformans and Histoplasma capsulatum but has not been reported in Candida isolates. Accumulation of obtusifolione correlated with nearly complete growth inhibition in these azole-resistant strains compared to that found in the susceptible parent strain, although the onset of growth inhibition only occurred at higher concentrations of itraconazole. ERG25 and ERG26 are the only genes assigned to the 4-demethylation process, of which the 3-ketoreductase is part. To verify whether mutations in these ERG25 genes contributed to obtusifolione accumulation, their nucleotide sequences were determined in all three related isolates. No mutations in ERG25 alleles of isolates C48 and C56 were found, suggesting that this gene is not involved in obtusifolione accumulation. The molecular basis for the accumulation of this sterol in these two strains remains to be established.
Figures
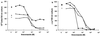
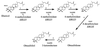
References
-
- Bodey G P. Resistance to antimicrobial agents revisited. Curr Opin Infect Dis. 1997;10:419–421.
-
- De Muri G P, Hostetter M K. Resistance to antifungal agents. Pediatr Clin N Am. 1995;42:665–685. - PubMed
-
- Denning D W, Baily G G, Hood S V. Azole resistance in Candida. Eur J Clin Microbiol Infect Dis. 1997;16:261–280. - PubMed
-
- Doignon F, Aigle M, Ribereau-Gayon P. Resistance to imidazoles and triazoles in Saccharomyces cerevisiae as a new dominant marker. Plasmid. 1993;30:224–233. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

