Airway epithelial CFTR mRNA expression in cystic fibrosis patients after repetitive administration of a recombinant adenovirus
- PMID: 10545523
- PMCID: PMC409826
- DOI: 10.1172/JCI7935
Airway epithelial CFTR mRNA expression in cystic fibrosis patients after repetitive administration of a recombinant adenovirus
Abstract
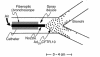
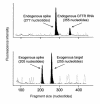
We sought to evaluate the ability of an E1(-), E3(-) adenovirus (Ad) vector (Ad(GV)CFTR.10) to transfer the normal human cystic fibrosis transmembrane conductance regulator (CFTR) cDNA to the airway epithelium of individuals with cystic fibrosis (CF). We administered Ad(GV)CFTR.10 at doses of 3 x 10(6) to 2 x 10(9) plaque-forming units over 9 months by endobronchial spray to 7 pairs of individuals with CF. Each 3-month cycle, we measured vector-derived versus endogenous CFTR mRNA in airway epithelial cells prior to therapy, as well as 3 and 30 days after therapy. The data demonstrate that (a) this strategy appears to be safe; (b) after the first administration, vector-derived CFTR cDNA expression in the CF airway epithelium is dose-dependent, with greater than 5% endogenous CFTR mRNA levels at the higher vector doses; (c) expression is transient, lasting less than 30 days; (d) expression can be achieved with a second administration, but only at intermediate doses, and no expression is observed with the third administration; and (e) the progressive lack of expression with repetitive administration does not closely correlate with induction of systemic anti-Ad neutralizing antibodies. The major advantage of an Ad vector is that it can deliver sufficient levels of CFTR cDNA to the airway epithelium so that CFTR expression protects the lungs from the respiratory manifestations of CF. However, this impressive level of expression is linked to the challenging fact that expression is limited in time. Although this can be initially overcome by repetitive administration, unknown mechanisms eventually limit this strategy, and further repetitive administration does not lead to repetitive expression.
Figures





Comment in
-
Gene transfer for cystic fibrosis.J Clin Invest. 1999 Nov;104(9):1165-6. doi: 10.1172/JCI8634. J Clin Invest. 1999. PMID: 10545512 Free PMC article. Review. No abstract available.
Similar articles
-
Aerosol and lobar administration of a recombinant adenovirus to individuals with cystic fibrosis. I. Methods, safety, and clinical implications.Hum Gene Ther. 2001 Jul 20;12(11):1369-82. doi: 10.1089/104303401750298535. Hum Gene Ther. 2001. PMID: 11485629 Clinical Trial.
-
[Aerosol administration of a replication defective recombinant adenovirus expressing normal human cDNA-CFTR in the respiratory tractus in patients with cystic fibrosis].C R Seances Soc Biol Fil. 1996;190(1):109-42. C R Seances Soc Biol Fil. 1996. PMID: 8881273 Clinical Trial. French.
-
Gene therapy for the respiratory manifestations of cystic fibrosis.Am J Respir Crit Care Med. 1995 Mar;151(3 Pt 2):S75-87. doi: 10.1164/ajrccm/151.3_Pt_2.S75. Am J Respir Crit Care Med. 1995. PMID: 7533609 Review.
-
Vector-specific complementation profiles of two independent primary defects in cystic fibrosis airways.Hum Gene Ther. 1998 Mar 20;9(5):635-48. doi: 10.1089/hum.1998.9.5-635. Hum Gene Ther. 1998. PMID: 9551612
-
Gene therapy in cystic fibrosis.Chest. 2001 Sep;120(3 Suppl):124S-131S. doi: 10.1378/chest.120.3_suppl.124s. Chest. 2001. PMID: 11555567 Review.
Cited by
-
The challenge of using gene- or cell-based therapies to treat lung disease.Mol Ther. 2012 Jun;20(6):1077-8. doi: 10.1038/mt.2012.91. Mol Ther. 2012. PMID: 22652992 Free PMC article. No abstract available.
-
Lung gene therapy-How to capture illumination from the light already present in the tunnel.Genes Dis. 2014 Sep;1(1):40-52. doi: 10.1016/j.gendis.2014.06.001. Genes Dis. 2014. PMID: 26161434 Free PMC article.
-
Disrupted adenovirus-based vaccines against small addictive molecules circumvent anti-adenovirus immunity.Hum Gene Ther. 2013 Jan;24(1):58-66. doi: 10.1089/hum.2012.163. Epub 2012 Dec 21. Hum Gene Ther. 2013. PMID: 23140508 Free PMC article.
-
Protection against P. aeruginosa with an adenovirus vector containing an OprF epitope in the capsid.J Clin Invest. 2005 May;115(5):1281-9. doi: 10.1172/JCI23135. Epub 2005 Apr 1. J Clin Invest. 2005. PMID: 15841217 Free PMC article.
-
Adenovirus: the first effective in vivo gene delivery vector.Hum Gene Ther. 2014 Jan;25(1):3-11. doi: 10.1089/hum.2013.2527. Hum Gene Ther. 2014. PMID: 24444179 Free PMC article. Review. No abstract available.
References
-
- Kerem B, et al. Identification of the cystic fibrosis gene: genetic analysis. Science. 1989;245:1073–1080. - PubMed
-
- Riordan JR, et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science. 1989;245:1066–1073. - PubMed
-
- Rommens JM, et al. Identification of the cystic fibrosis gene: chromosome walking and jumping. Science. 1989;245:1059–1065. - PubMed
-
- Welsh, M.J., Tsui, L.-C., Boat, T.F., and Beaudet, A.L. 1995. Cystic fibrosis. In The metabolic and molecular basis of inherited disease. C.R. Scriver, A.L. Beaudet, W.S. Sly, and D. Valle, editors. McGraw-Hill. New York, NY. 3799–3876.
-
- Cystic Fibrosis Foundation. 1997. Opening doors to opportunities. New challenges and new interventions. 1997 Annual Report Cystic Fibrosis Foundation. Bethesda, MD. 1–13.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

