The role of protease-activated receptor-2 (PAR2) in the modulation of beating of the mouse isolated ureter: lack of involvement of mast cells or sensory nerves
- PMID: 10556919
- PMCID: PMC1571711
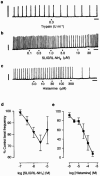
- DOI: 10.1038/sj.bjp.0702871
The role of protease-activated receptor-2 (PAR2) in the modulation of beating of the mouse isolated ureter: lack of involvement of mast cells or sensory nerves
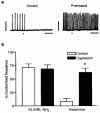
Abstract
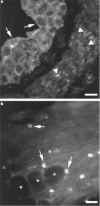
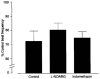
1 The localization of protease-activated receptor-2 (PAR2) and the effects of PAR2 activators were investigated in the mouse isolated ureter in order to test the hypothesis that PAR2 activation may initiate neuropeptide release from sensory nerve fibres and hence contribute to inflammation. 2 PAR2 was localized by fluorescence immunohistochemistry to both the smooth muscle and epithelium of the ureter. Macrophage-like cells in the adventitia of the ureter were also PAR2-immunoreactive. PAR2-immunoreactivity was not observed in mast cells or nerve fibres. 3 In circular muscle preparations of the ureter in which continuous rhythmic beating was induced by KCl (20 mM) and the thromboxane A2 mimetic U46619 (0.3 microM), trypsin (0.3 U ml-1) reduced beat frequency to 84.6+/-2.0% of control rates. The PAR2-selective peptide agonist SLIGRL-NH2 concentration-dependently (0.1-3.0 microM) slowed beat frequency to a maximum of 72.7+/-2.0%. 4 Histamine (1-300 microM) was more efficacious than SLIGRL-NH2 in inhibiting ureter beat frequency in a concentration-dependent manner to a maximum (at 300 microM) of 7.9+/-2.5% of the control rate. 5 Pretreatment of preparations with capsaicin (10 microM for 30 min) markedly attenuated the inhibitory effect of histamine, but not that of SLIGRL-NH2, indicating a role for sensory nerves in the inhibitory effect of histamine only. 6 The inhibitory effect of SLIGRL-NH2 on ureter beat frequency was unaffected by the nitric oxide (NO) synthase inhibitor, L-NOARG (100 microM) or the cyclo-oxygenase inhibitor, indomethacin (3 microM). 7 In conclusion, PAR2 activation causes inhibition of beating in the mouse ureter that is not mediated by axon reflex release of inhibitory neuropeptides. This inhibitory effect of PAR2 appears to be mediated directly on smooth muscle cells, although the contribution of non-NO, non-prostanoid epithelium-derived factors cannot be ruled out.
Figures
Similar articles
-
Effect of protease-activated receptor (PAR)-1, -2 and -4-activating peptides, thrombin and trypsin in rat isolated airways.Br J Pharmacol. 2000 Dec;131(8):1584-91. doi: 10.1038/sj.bjp.0703738. Br J Pharmacol. 2000. PMID: 11139435 Free PMC article.
-
Trypsin-induced, neurokinin-mediated contraction of guinea pig bronchus.Am J Respir Crit Care Med. 2000 Nov;162(5):1662-7. doi: 10.1164/ajrccm.162.5.9912099. Am J Respir Crit Care Med. 2000. PMID: 11069793
-
Proteinase-activated receptor-2 (PAR2): vascular effects of a PAR2-derived activating peptide via a receptor different than PAR2.J Pharmacol Exp Ther. 2002 Dec;303(3):985-92. doi: 10.1124/jpet.102.040352. J Pharmacol Exp Ther. 2002. PMID: 12438518
-
Role of PGE(2) in protease-activated receptor-1, -2 and -4 mediated relaxation in the mouse isolated trachea.Br J Pharmacol. 2001 Jan;132(1):93-100. doi: 10.1038/sj.bjp.0703776. Br J Pharmacol. 2001. PMID: 11156565 Free PMC article.
-
Heterogeneity of neuronal and smooth muscle receptors involved in the VIP- and PACAP-induced relaxations of the pig intravesical ureter.Br J Pharmacol. 2004 Jan;141(1):123-31. doi: 10.1038/sj.bjp.0705582. Epub 2003 Dec 8. Br J Pharmacol. 2004. PMID: 14662737 Free PMC article.
Cited by
-
Protease-activated receptors in health and disease.Physiol Rev. 2023 Jan 1;103(1):717-785. doi: 10.1152/physrev.00044.2021. Epub 2022 Jul 28. Physiol Rev. 2023. PMID: 35901239 Free PMC article. Review.
-
Differential expression of protease-activated receptors-1 and -2 in stromal fibroblasts of normal, benign, and malignant human tissues.Am J Pathol. 2001 Jun;158(6):2031-41. doi: 10.1016/S0002-9440(10)64675-5. Am J Pathol. 2001. PMID: 11395381 Free PMC article.
References
-
- BÖHM S.K., MCCONALOGUE K., KONG W., BUNNETT N.W. Proteinase-activated receptors: new functions for old enzymes. News Physiol. Sci. 1998;13:231–240. - PubMed
-
- COCKS T.M., FONG B., CHOW J.M., ANDERSON G.P., FRAUMAN A.G., GOLDIE R.G., HENRY P.J., CARR M.J., HAMILTON J.R., MOFFATT J.D. A protective role for protease-activated receptors in the airways. Nature. 1999a;398:156–160. - PubMed
-
- COCKS T.M., SOZZI V., MOFFATT J.D., SELEMIDIS S. Protease-activated receptors mediate apamin-sensitive relaxation of mouse and guinea pig gastrointestinal smooth muscle. Gastroenterology. 1999b;116:586–592. - PubMed
-
- D'ANDREA M.R., DERIAN C.K., LETURCQ D., BAKER S.M., BRUNMARK A., LING P., DARROW A.L., SANTULLI R.J., BRASS L.F., ANDRADE-GORDON P. Characterization of protease-activated receptor-2 immunoreactivity in normal human tissues. J. Histochem. Cytochem. 1998;46:157–164. - PubMed
-
- DÉRY O., CORVERA C.U., STEINHOFF M., BUNNETT N.W. Proteinase-activated receptors: novel mechanisms of signalling by serine proteases. Am. J. Physiol. 1998;274:C1429–C1542. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

