Farnesol is utilized for isoprenoid biosynthesis in plant cells via farnesyl pyrophosphate formed by successive monophosphorylation reactions
- PMID: 10557276
- PMCID: PMC23903
- DOI: 10.1073/pnas.96.23.13080
Farnesol is utilized for isoprenoid biosynthesis in plant cells via farnesyl pyrophosphate formed by successive monophosphorylation reactions
Abstract
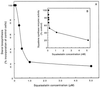
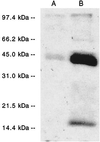
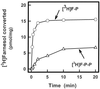
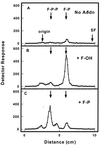
The ability of Nicotiana tabacum cell cultures to utilize farnesol (F-OH) for sterol and sesquiterpene biosynthesis was investigated. [(3)H]F-OH was readily incorporated into sterols by rapidly growing cell cultures. However, the incorporation rate into sterols was reduced by greater than 70% in elicitor-treated cell cultures whereas a substantial proportion of the radioactivity was redirected into capsidiol, an extracellular sesquiterpene phytoalexin. The incorporation of [(3)H]F-OH into sterols was inhibited by squalestatin 1, suggesting that [(3)H]F-OH was incorporated via farnesyl pyrophosphate (F-P-P). Consistent with this possibility, N. tabacum proteins were metabolically labeled with [(3)H]F-OH or [(3)H]geranylgeraniol ([(3)H]GG-OH). Kinase activities converting F-OH to farnesyl monophosphate (F-P) and, subsequently, F-P-P were demonstrated directly by in vitro enzymatic studies. [(3)H]F-P and [(3)H]F-P-P were synthesized when exogenous [(3)H]F-OH was incubated with microsomal fractions and CTP. The kinetics of formation suggested a precursor-product relationship between [(3)H]F-P and [(3)H]F-P-P. In agreement with this kinetic pattern of labeling, [(32)P]F-P and [(32)P]F-P-P were synthesized when microsomal fractions were incubated with F-OH and F-P, respectively, with [gamma-(32)P]CTP serving as the phosphoryl donor. Under similar conditions, the microsomal fractions catalyzed the enzymatic conversion of [(3)H]GG-OH to [(3)H]geranylgeranyl monophosphate and [(3)H]geranylgeranyl pyrophosphate ([(3)H]GG-P-P) in CTP-dependent reactions. A novel biosynthetic mechanism involving two successive monophosphorylation reactions was supported by the observation that [(3)H]CTP was formed when microsomes were incubated with [(3)H]CDP and either F-P-P or GG-P-P, but not F-P. These results document the presence of at least two CTP-mediated kinases that provide a mechanism for the utilization of F-OH and GG-OH for the biosynthesis of isoprenoid lipids and protein isoprenylation.
Figures
Similar articles
-
Farnesol is utilized for protein isoprenylation and the biosynthesis of cholesterol in mammalian cells.Biochem Biophys Res Commun. 1995 Jun 15;211(2):590-9. doi: 10.1006/bbrc.1995.1854. Biochem Biophys Res Commun. 1995. PMID: 7794274
-
Phosphorylation of farnesol in rat liver microsomes: properties of farnesol kinase and farnesyl phosphate kinase.Arch Biochem Biophys. 1998 May 15;353(2):191-8. doi: 10.1006/abbi.1998.0611. Arch Biochem Biophys. 1998. PMID: 9606952
-
Geranylgeraniol overcomes the block of cell proliferation by lovastatin in C6 glioma cells.J Neurochem. 1998 Jun;70(6):2397-405. doi: 10.1046/j.1471-4159.1998.70062397.x. J Neurochem. 1998. PMID: 9603204
-
Sterols and isoprenoids: signaling molecules derived from the cholesterol biosynthetic pathway.Annu Rev Biochem. 1999;68:157-85. doi: 10.1146/annurev.biochem.68.1.157. Annu Rev Biochem. 1999. PMID: 10872447 Review.
-
Organization of isoprenoid biosynthesis.Biochem Soc Trans. 1996 Aug;24(3):677-82. doi: 10.1042/bst0240677. Biochem Soc Trans. 1996. PMID: 8878825 Review. No abstract available.
Cited by
-
Farnesol-induced cell death and stimulation of 3-hydroxy-3-methylglutaryl-coenzyme A reductase activity in tobacco cv bright yellow-2 cells.Plant Physiol. 2000 Aug;123(4):1257-68. doi: 10.1104/pp.123.4.1257. Plant Physiol. 2000. PMID: 10938345 Free PMC article.
-
Isolation and functional expression of human COQ2, a gene encoding a polyprenyl transferase involved in the synthesis of CoQ.Biochem J. 2004 Sep 1;382(Pt 2):519-26. doi: 10.1042/BJ20040261. Biochem J. 2004. PMID: 15153069 Free PMC article.
-
The plastidial 2-C-methyl-D-erythritol 4-phosphate pathway provides the isoprenyl moiety for protein geranylgeranylation in tobacco BY-2 cells.Plant Cell. 2009 Jan;21(1):285-300. doi: 10.1105/tpc.108.063248. Epub 2009 Jan 9. Plant Cell. 2009. PMID: 19136647 Free PMC article.
-
Identification of a novel abscisic acid-regulated farnesol dehydrogenase from Arabidopsis.Plant Physiol. 2010 Nov;154(3):1116-27. doi: 10.1104/pp.110.157784. Epub 2010 Aug 31. Plant Physiol. 2010. PMID: 20807998 Free PMC article.
-
Role of Phosphatidic Acid Phosphatase Domain Containing 2 in Squalestatin 1-Mediated Activation of the Constitutive Androstane Receptor in Primary Cultured Rat Hepatocytes.Drug Metab Dispos. 2016 Mar;44(3):352-5. doi: 10.1124/dmd.115.068437. Epub 2015 Dec 23. Drug Metab Dispos. 2016. PMID: 26700959 Free PMC article.
References
-
- Grunler J, Ericsson J, Dallner G. Biochim Biophys Acta. 1994;1212:259–277. - PubMed
-
- Goldstein J L, Brown M S. Nature (London) 1990;343:425–430. - PubMed
-
- Jiang G, McKenzie T L, Conrad D G, Shechter I. J Biol Chem. 1993;268:12818–12824. - PubMed
-
- Bostock R M, Kuc J, Laine R A. Science. 1981;212:67–69. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

