Differential methylation of Epstein-Barr virus latency promoters facilitates viral persistence in healthy seropositive individuals
- PMID: 10559309
- PMCID: PMC113046
- DOI: 10.1128/JVI.73.12.9959-9968.1999
Differential methylation of Epstein-Barr virus latency promoters facilitates viral persistence in healthy seropositive individuals
Abstract
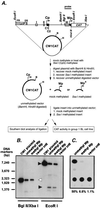
Epstein-Barr virus (EBV) establishes a life-long infection in humans, with distinct viral latency programs predominating during acute and chronic phases of infection. Only a subset of the EBV latency-associated antigens present during the acute phase of EBV infection are expressed in the latently infected memory B cells that serve as the long-term EBV reservoir. Since the EBV immortalization program elicits a potent cellular immune response, downregulation of viral gene expression in the long-term latency reservoir is likely to facilitate evasion of the immune response and persistence of EBV in the immunocompetent host. Tissue culture and tumor models of restricted EBV latency have consistently demonstrated a critical role for methylation of the viral genome in maintaining the restricted pattern of latency-associated gene expression. Here we extend these observations to demonstrate that the EBV genomes in the memory B-cell reservoir are also heavily and discretely methylated. This analysis reveals that methylation of the viral genome is a normal aspect of EBV infection in healthy immunocompetent individuals and is not restricted to the development of EBV-associated tumors. In addition, the pattern of methylation very likely accounts for the observed inhibition of the EBV immortalization program and the establishment and maintenance of a restricted latency program. Thus, EBV appears to be the first example of a parasite that usurps the host cell-directed methylation system to regulate pathogen gene expression and thereby establish a chronic infection.
Figures


References
-
- Babcock G J, Decker L L, Volk M, Thorley-Lawson D A. EBV persistence in memory B cells in vivo. Immunity. 1998;9:395–404. - PubMed
-
- Barlow D P. Methylation and imprinting: from host defense to gene regulation? Science. 1993;260:309–310. - PubMed
-
- Bender J. Cytosine methylation of repeated sequences in eukaryotes: the role of DNA pairing. Trends Biochem Sci. 1998;23:252–256. - PubMed
-
- Bestor T H. Gene silencing. Methylation meets acetylation. Nature. 1998;393:311–312. . (News; comment.) - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

