Cathepsin S controls the trafficking and maturation of MHC class II molecules in dendritic cells
- PMID: 10562280
- PMCID: PMC2156161
- DOI: 10.1083/jcb.147.4.775
Cathepsin S controls the trafficking and maturation of MHC class II molecules in dendritic cells
Abstract
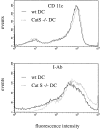
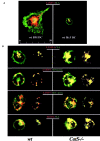
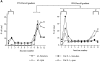
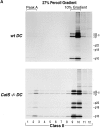
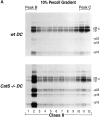
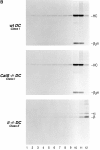
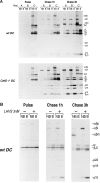
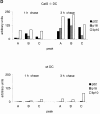
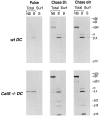
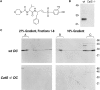
Before a class II molecule can be loaded with antigenic material and reach the surface to engage CD4+ T cells, its chaperone, the class II-associated invariant chain (Ii), is degraded in a stepwise fashion by proteases in endocytic compartments. We have dissected the role of cathepsin S (CatS) in the trafficking and maturation of class II molecules by combining the use of dendritic cells (DC) from CatS(-/-) mice with a new active site-directed probe for direct visualization of active CatS. Our data demonstrate that CatS is active along the entire endocytic route, and that cleavage of the lysosomal sorting signal of Ii by CatS can occur there in mature DC. Genetic disruption of CatS dramatically reduces the flow of class II molecules to the cell surface. In CatS(-/-) DC, the bulk of major histocompatibility complex (MHC) class II molecules is retained in late endocytic compartments, although paradoxically, surface expression of class II is largely unaffected. The greatly diminished but continuous flow of class II molecules to the cell surface, in conjunction with their long half-life, can account for the latter observation. We conclude that in DC, CatS is a major determinant in the regulation of intracellular trafficking of MHC class II molecules.
Figures




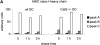
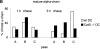

References
-
- Bakke O., Dobberstein B. MHC class II-associated invariant chain contains a sorting signal for endosomal compartments. Cell. 1990;63:707–716 . - PubMed
-
- Banchereau J., Steinman R.M. Dendritic cells and the control of immunity. Nature. 1998;392:245–252 . - PubMed
-
- Brown M.S., Goldstein J.L. The SREBP pathwayregulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell. 1997;89:331–340 . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

