Yes-associated protein 65 localizes p62(c-Yes) to the apical compartment of airway epithelia by association with EBP50
- PMID: 10562288
- PMCID: PMC2156157
- DOI: 10.1083/jcb.147.4.879
Yes-associated protein 65 localizes p62(c-Yes) to the apical compartment of airway epithelia by association with EBP50
Abstract
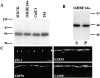
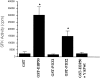
We recently showed that the COOH terminus of the cystic fibrosis transmembrane conductance regulator associates with the submembranous scaffolding protein EBP50 (ERM-binding phosphoprotein 50 kD; also called Na(+)/H(+) exchanger regulatory factor). Since EBP50 associates with ezrin, this interaction links the cystic fibrosis transmembrane conductance regulator (CFTR) to the cortical actin cytoskeleton. EBP50 has two PDZ domains, and CFTR binds with high affinity to the first PDZ domain. Here, we report that Yes-associated protein 65 (YAP65) binds with high affinity to the second EBP50 PDZ domain. YAP65 is concentrated at the apical membrane in airway epithelia and interacts with EBP50 in cells. The COOH terminus of YAP65 is necessary and sufficient to mediate association with EBP50. The EBP50-YAP65 interaction is involved in the compartmentalization of YAP65 at the apical membrane since mutant YAP65 proteins lacking the EBP50 interaction motif are mislocalized when expressed in airway epithelial cells. In addition, we show that the nonreceptor tyrosine kinase c-Yes is contained within EBP50 protein complexes by association with YAP65. Subapical EBP50 protein complexes, containing the nonreceptor tyrosine kinase c-Yes, may regulate apical signal transduction pathways leading to changes in ion transport, cytoskeletal organization, or gene expression in epithelial cells.
Figures

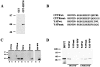


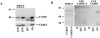




Similar articles
-
A PDZ-interacting domain in CFTR is an apical membrane polarization signal.J Clin Invest. 1999 Nov;104(10):1353-61. doi: 10.1172/JCI7453. J Clin Invest. 1999. PMID: 10562297 Free PMC article.
-
An apical PDZ protein anchors the cystic fibrosis transmembrane conductance regulator to the cytoskeleton.J Biol Chem. 1998 Jul 31;273(31):19797-801. doi: 10.1074/jbc.273.31.19797. J Biol Chem. 1998. PMID: 9677412
-
Identification of EBP50: A PDZ-containing phosphoprotein that associates with members of the ezrin-radixin-moesin family.J Cell Biol. 1997 Oct 6;139(1):169-79. doi: 10.1083/jcb.139.1.169. J Cell Biol. 1997. PMID: 9314537 Free PMC article.
-
CFTR-NHERF2-LPA₂ Complex in the Airway and Gut Epithelia.Int J Mol Sci. 2017 Sep 4;18(9):1896. doi: 10.3390/ijms18091896. Int J Mol Sci. 2017. PMID: 28869532 Free PMC article. Review.
-
Roles of NHERF1/EBP50 in cancer.Curr Mol Med. 2008 Sep;8(6):459-68. doi: 10.2174/156652408785748031. Curr Mol Med. 2008. PMID: 18781953 Review.
Cited by
-
The transmembrane protein CBP plays a role in transiently anchoring small clusters of Thy-1, a GPI-anchored protein, to the cytoskeleton.J Cell Sci. 2009 Nov 1;122(Pt 21):3966-72. doi: 10.1242/jcs.049346. Epub 2009 Oct 13. J Cell Sci. 2009. PMID: 19825940 Free PMC article.
-
Interactions of connexins with other membrane channels and transporters.Prog Biophys Mol Biol. 2007 May-Jun;94(1-2):233-44. doi: 10.1016/j.pbiomolbio.2007.03.002. Epub 2007 Mar 14. Prog Biophys Mol Biol. 2007. PMID: 17475311 Free PMC article. Review.
-
Aberrant nuclear localization of EBP50 promotes colorectal carcinogenesis in xenotransplanted mice by modulating TCF-1 and β-catenin interactions.J Clin Invest. 2012 May;122(5):1881-94. doi: 10.1172/JCI45661. Epub 2012 Apr 2. J Clin Invest. 2012. PMID: 22466651 Free PMC article.
-
A novel role of protein tyrosine kinase2 in mediating chloride secretion in human airway epithelial cells.PLoS One. 2011;6(7):e21991. doi: 10.1371/journal.pone.0021991. Epub 2011 Jul 13. PLoS One. 2011. PMID: 21765932 Free PMC article.
-
Targeted disruption of the mouse NHERF-1 gene promotes internalization of proximal tubule sodium-phosphate cotransporter type IIa and renal phosphate wasting.Proc Natl Acad Sci U S A. 2002 Aug 20;99(17):11470-5. doi: 10.1073/pnas.162232699. Epub 2002 Aug 8. Proc Natl Acad Sci U S A. 2002. PMID: 12169661 Free PMC article.
References
-
- Biemesderfer D., Rutherford P.A., Nagy T., Pizzonia J.H., Abu-Alfa A.K., Aronson P.S. Monoclonal antibodies for high-resolution localization of NHE3 in adult and neonatal rat kidney. Am. J. Physiol. 1997;273:289–299 . - PubMed
-
- Brant S.R., Yun C.H., Donowitz M., Tse C.M. Cloning, tissue distribution, and functional analysis of the human Na+/H+ exchanger isoform, NHE3. Am. J. Physiol. 1995;269:198–206 . - PubMed
-
- Brenman J.E., Chao D.S., Gee S.H., McGee A.W., Craven S.E., Santillano D.R., Wu Z., Huang F., Xia H., Peters M.F., Froehner S.C., Bredt D.S. Interaction of nitric oxide synthase with the postsynaptic density protein PSD-95 and α1-syntrophin mediated by PDZ domains. Cell. 1996;84:757–767 . - PubMed
-
- Chen H.I., Einbond A., Kwak S.J., Linn H., Koepf E., Peterson S., Kelly J.W., Sudol M. Characterization of the WW domain of human Yes-associated protein and its polyproline-containing ligands. J. Biol. Chem. 1997;272:17070–17077 . - PubMed

