Regulation of smooth muscle cell migration and integrin expression by the Gax transcription factor
- PMID: 10562309
- PMCID: PMC409839
- DOI: 10.1172/JCI7251
Regulation of smooth muscle cell migration and integrin expression by the Gax transcription factor
Abstract
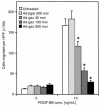
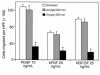
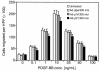
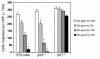
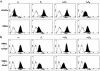
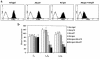
Homeobox transcription factors specify body plan by regulating differentiation, proliferation, and migration at a cellular level. The homeobox transcription factor Gax is expressed in quiescent vascular smooth muscle cells (VSMCs), and its expression is downregulated by vascular injury or other conditions that lead to VSMC proliferation. Previous investigations demonstrate that Gax may regulate VSMC proliferation by upregulating the cyclin-dependent kinase (cdk) inhibitor p21. Here we examined whether Gax influences VSMC migration, a key feature in the development of stenotic lesions after balloon injury. Transduction of a Gax cDNA inhibited the migratory response of VSMCs toward PDGF-BB, basic fibroblast growth factor, or hepatocyte growth factor/scatter factor. Gax expression also inhibited migration of NIH.3T3 fibroblasts and embryonic fibroblasts lacking p53. Gax was unable to inhibit the migration of fibroblasts lacking p21, but this effect could be restored in these cells by providing exogenous p21 or by overexpressing another cdk inhibitor, p16. Flow cytometric analysis implicated a Gax-mediated downregulation of alpha(v)beta(3) and alpha(v)beta(5) integrin expression in VSMCs as a potential cause for reduced cell motility. Gax specifically downregulated beta(3) and beta(5) in VSMCs in culture and after acute vascular injury in vivo. Repression of integrin expression was also found in NIH 3T3 cells and p53 knockout fibroblasts, but not in p21-knockout fibroblasts, unless these cells express exogenous p21 or p16. These data suggest that cycle progression, integrin expression, and cell migration can be regulated in VSMCs by the homeobox gene product Gax.
Figures


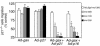


Similar articles
-
Role of JNK, p38, and ERK in platelet-derived growth factor-induced vascular proliferation, migration, and gene expression.Arterioscler Thromb Vasc Biol. 2003 May 1;23(5):795-801. doi: 10.1161/01.ATV.0000066132.32063.F2. Epub 2003 Mar 13. Arterioscler Thromb Vasc Biol. 2003. PMID: 12637337
-
Bax-mediated cell death by the Gax homeoprotein requires mitogen activation but is independent of cell cycle activity.EMBO J. 1998 Jul 1;17(13):3576-86. doi: 10.1093/emboj/17.13.3576. EMBO J. 1998. PMID: 9649428 Free PMC article.
-
Phosphoinositide-dependent kinase 1 and p21-activated protein kinase mediate reactive oxygen species-dependent regulation of platelet-derived growth factor-induced smooth muscle cell migration.Circ Res. 2004 May 14;94(9):1219-26. doi: 10.1161/01.RES.0000126848.54740.4A. Epub 2004 Apr 1. Circ Res. 2004. PMID: 15059930
-
The role of homeobox genes in vascular remodeling and angiogenesis.Circ Res. 2000 Nov 10;87(10):865-72. doi: 10.1161/01.res.87.10.865. Circ Res. 2000. PMID: 11073881 Review.
-
Tissue factor regulation in vascular smooth muscle: a summary of studies performed using in vivo and in vitro models.Am J Cardiol. 1993 Sep 9;72(8):55C-60C. doi: 10.1016/0002-9149(93)90256-c. Am J Cardiol. 1993. PMID: 8372802 Review.
Cited by
-
Integrin beta3 regulates clonality and fate of smooth muscle-derived atherosclerotic plaque cells.Nat Commun. 2018 May 25;9(1):2073. doi: 10.1038/s41467-018-04447-7. Nat Commun. 2018. PMID: 29802249 Free PMC article.
-
Inhibition of integrin alpha v/beta 5 mitigates the protective effect induced by irisin in hemorrhage.Exp Mol Pathol. 2023 Dec;134:104869. doi: 10.1016/j.yexmp.2023.104869. Epub 2023 Sep 27. Exp Mol Pathol. 2023. PMID: 37690529 Free PMC article.
-
Analysis of homeobox gene action may reveal novel angiogenic pathways in normal placental vasculature and in clinical pregnancy disorders associated with abnormal placental angiogenesis.Front Pharmacol. 2014 Jun 4;5:133. doi: 10.3389/fphar.2014.00133. eCollection 2014. Front Pharmacol. 2014. PMID: 24926269 Free PMC article. Review.
-
miR-301a is a candidate oncogene that targets the homeobox gene Gax in human hepatocellular carcinoma.Dig Dis Sci. 2012 May;57(5):1171-80. doi: 10.1007/s10620-012-2099-2. Epub 2012 Feb 29. Dig Dis Sci. 2012. PMID: 22373864
-
Dietary potassium regulates vascular calcification and arterial stiffness.JCI Insight. 2017 Oct 5;2(19):e94920. doi: 10.1172/jci.insight.94920. JCI Insight. 2017. PMID: 28978809 Free PMC article.
References
-
- Niehrs C, Keller R, Cho KWY, De Robertis EM. The homeobox gene goosecoid controls cell migration in Xenopus embryos. Cell. 1993;72:491–503. - PubMed
-
- Salser SJ, Kenyon C. Activation of a C. elegans Antennapedia homologue in migrating cells controls their direction of migration. Nature. 1992;355:255–258. - PubMed
-
- Gorski DH, Patel CV, Walsh K. Homeobox transcription factor regulation in the cardiovascular system. Trends Cardiovasc Med. 1993;3:184–190. - PubMed
-
- Olson EN, Rosenthal N. Homeobox genes and muscle patterning. Cell. 1994;79:9–12. - PubMed
-
- Scott MP, Tamkun JW, Hartzell GW. The structure and function of the homeodomain. Biochim Biophys Acta. 1989;989:25–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

