Oxidative damage and age-related macular degeneration
- PMID: 10562656
- PMCID: PMC1773059
Oxidative damage and age-related macular degeneration
Abstract
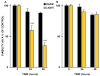
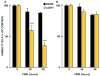
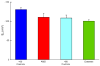
This article provides current information on the potential role of oxidation in relation to age-related macular degeneration (AMD). The emphasis is placed on the generation of oxidants and free radicals and the protective effects of antioxidants in the outer retina, with specific emphasis on the photoreceptor cells, the retinal pigment epithelium and the choriocapillaris. The starting points include a discussion and a definition of what radicals are, their endogenous sources, how they react, and what damage they may cause. The photoreceptor/pigment epithelium complex is exposed to sunlight, is bathed in a near-arterial level of oxygen, and membranes in this complex contain high concentrations of polyunsaturated fatty acids, all considered to be potential factors leading to oxidative damage. Actions of antioxidants such as glutathione, vitamin C, superoxide dismutase, catalase, vitamin E and the carotenoids are discussed in terms of their mechanisms of preventing oxidative damage. The phototoxicity of lipofuscin, a group of complex autofluorescent lipid/protein aggregates that accumulate in the retinal pigment epithelium, is described and evidence is presented suggesting that intracellular lipofuscin is toxic to these cells, thus supporting a role for lipofuscin in aging and AMD. The theory that AMD is primarily due to a photosensitizing injury to the choriocapillaris is evaluated. Results are presented showing that when protoporphyric mice are exposed to blue light there is an induction in the synthesis of Type IV collagen synthesis by the choriocapillary endothelium, which leads to a thickened Bruch's membrane and to the appearance of sub-retinal pigment epithelial fibrillogranular deposits, which are similar to basal laminar deposits. The hypothesis that AMD may result from oxidative injury to the retinal pigment epithelium is further evaluated in experiments designed to test the protective effects of glutathione in preventing damage to cultured human pigment epithelial cells exposed to an oxidant. Experiments designed to increase the concentration of glutathione in pigment epithelial cells using dimethylfumarate, a monofunctional inducer, are described in relation to the ability of these cells to survive an oxidative challenge. While all these models provide undisputed evidence of oxidative damage to the retinal pigment epithelium and the choriocapillaris that is both light- and oxygen-dependent, it nevertheless is still unclear at this time what the precise linkage is between oxidation-induced events and the onset and progression of AMD.
Figures



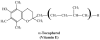






References
-
- Young RW. Pathophysiology of age-related macular degeneration. Surv Ophthalmol. 1987;31:291–306. - PubMed
-
- Bressler NM, Bressler SB, Fine SL. Age-related macular degeneration. Surv Ophthalmol. 1988;32:375–413. - PubMed
-
- Noell WK, Walker VS, Kang BS, Berman S. Retinal damage by light in rats. Invest Ophthalmol. 1966;5:450–73. - PubMed
-
- Young RW. Solar radiation and age-related macular degeneration. Surv Ophthalmol. 1988;32:252–69. - PubMed
-
- Boulton ME. Ageing of the retinal pigment epithelium. Prog Retin Eye Res. 1991;11:125–51.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

