The AF1 and AF2 domains of the androgen receptor interact with distinct regions of SRC1
- PMID: 10567563
- PMCID: PMC84931
- DOI: 10.1128/MCB.19.12.8383
The AF1 and AF2 domains of the androgen receptor interact with distinct regions of SRC1
Abstract
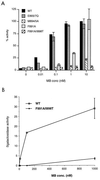
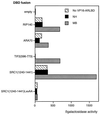
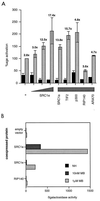
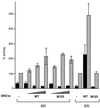
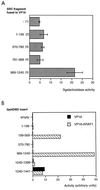
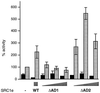
The androgen receptor is unusual among nuclear receptors in that most, if not all, of its activity is mediated via the constitutive activation function in the N terminus. Here we demonstrate that p160 coactivators such as SRC1 (steroid receptor coactivator 1) interact directly with the N terminus in a ligand-independent manner via a conserved glutamine-rich region between residues 1053 and 1123. Although SRC1 is capable of interacting with the ligand-binding domain by means of LXXLL motifs, this interaction is not essential since an SRC1 mutant with no functional LXXLL motifs retains its ability to potentiate androgen receptor activity. In contrast, mutants lacking the glutamine-rich region are inactive, indicating that this region is both necessary and sufficient for recruitment of SRC1 to the androgen receptor. This recruitment is in direct contrast to the recruitment of SRC1 to the estrogen receptor, which requires interaction with the ligand-binding domain.
Figures

References
-
- Alen P, Claessens F, Schoenmakers E, Swinnen J V, Verhoeven G, Rombauts W, Peeters B. Interaction of the putative androgen receptor-specific coactivator ARA70/ELE1a with multiple steroid receptors and identification of an internally deleted ELE1β isoform. Mol Endocrinol. 1999;13:117–128. - PubMed
-
- Anzick S L, Kononen J, Walker R L, Azorsa D O, Tanner M M, Guan X-Y, Sauter G, Kallioniemi O-P, Trent J M, Meltzer P S. AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science. 1997;277:965–968. - PubMed
-
- Bannister A J, Kouzarides T. The CBP co-activator is a histone acetyltransferase. Nature. 1996;1996:641–643. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
