Activation of presynaptic cAMP-dependent protein kinase is required for induction of cerebellar long-term potentiation
- PMID: 10575019
- PMCID: PMC6782423
- DOI: 10.1523/JNEUROSCI.19-23-10221.1999
Activation of presynaptic cAMP-dependent protein kinase is required for induction of cerebellar long-term potentiation
Abstract
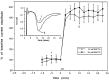
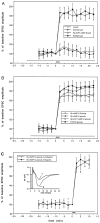
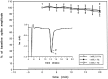
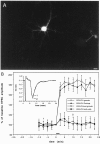
Cerebellar long-term potentiation (LTP) is a persistent increase in the strength of the granule cell-Purkinje neuron synapse that occurs after brief stimulation of granule cell axons at 2-8 Hz. Previous work has indicated that cerebellar LTP induction requires presynaptic Ca influx, stimulation of Ca-sensitive adenylyl cyclase, and activation of PKA. The evidence implicating PKA has come from bath application of drugs during LTP induction, an approach that does not distinguish between PKA activation in the presynaptic or postsynaptic cell. Although bath application of PKA inhibitor drugs (KT5720, Rp-8CPT-cAMP-S) blocked LTP induction in granule cell-Purkinje neuron pairs in culture, selective application to granule cell or Purkinje neuron somata via patch pipettes did not. We hypothesized that presynaptic PKA activation is required for LTP induction but that drugs applied to the granule cell soma cannot diffuse to the terminal within this timescale. To test this hypothesis, we transfected cerebellar cultures with an expression vector encoding a peptide inhibitor of PKA [Rous sarcoma virus (RSV)-protein kinase A inhibitor (PKI)]. Transfection of RSV-PKI into presynaptic granule cells, but not postsynaptic Purkinje neurons or glial cells, blocked LTP induction produced by either synaptic stimulation or an exogenous cAMP analog. An expression vector encoding a control peptide with no PKA inhibitory activity was ineffective. These results show that induction of cerebellar LTP requires a presynaptic signaling cascade, including Ca influx, stimulation of Ca-sensitive adenylyl cyclase, and activation of PKA, and argue against a requirement for postsynaptic Ca signals or their sequelae.
Figures

Similar articles
-
β-Adrenergic Receptors/Epac Signaling Increases the Size of the Readily Releasable Pool of Synaptic Vesicles Required for Parallel Fiber LTP.J Neurosci. 2020 Nov 4;40(45):8604-8617. doi: 10.1523/JNEUROSCI.0716-20.2020. Epub 2020 Oct 12. J Neurosci. 2020. PMID: 33046543 Free PMC article.
-
Synaptically evoked glutamate transport currents may be used to detect the expression of long-term potentiation in cerebellar culture.J Neurophysiol. 1998 Jun;79(6):3151-6. doi: 10.1152/jn.1998.79.6.3151. J Neurophysiol. 1998. PMID: 9636115
-
Cyclic AMP mediates a presynaptic form of LTP at cerebellar parallel fiber synapses.Neuron. 1996 Apr;16(4):797-803. doi: 10.1016/s0896-6273(00)80099-9. Neuron. 1996. PMID: 8607997
-
On the induction of postsynaptic granule cell-Purkinje neuron LTP and LTD.Cerebellum. 2010 Sep;9(3):284-90. doi: 10.1007/s12311-010-0174-9. Cerebellum. 2010. PMID: 20446074 Free PMC article. Review.
-
Nitric oxide and carbon monoxide as possible retrograde messengers in hippocampal long-term potentiation.J Neurobiol. 1994 Jun;25(6):652-65. doi: 10.1002/neu.480250607. J Neurobiol. 1994. PMID: 8071665 Review.
Cited by
-
A new form of cerebellar long-term potentiation is postsynaptic and depends on nitric oxide but not cAMP.Proc Natl Acad Sci U S A. 2002 Jun 11;99(12):8389-93. doi: 10.1073/pnas.122206399. Epub 2002 Jun 4. Proc Natl Acad Sci U S A. 2002. PMID: 12048250 Free PMC article.
-
Presynaptically expressed long-term depression at cerebellar parallel fiber synapses.Pflugers Arch. 2009 Feb;457(4):865-75. doi: 10.1007/s00424-008-0555-9. Epub 2008 Jul 29. Pflugers Arch. 2009. PMID: 18663469
-
Differential distribution of exchange proteins directly activated by cyclic AMP within the adult rat retina.Neuroscience. 2010 Feb 3;165(3):955-67. doi: 10.1016/j.neuroscience.2009.10.054. Epub 2009 Oct 31. Neuroscience. 2010. PMID: 19883736 Free PMC article.
-
Modulation of neurotransmitter release by the second messenger-activated protein kinases: implications for presynaptic plasticity.Pharmacol Ther. 2005 Jan;105(1):69-84. doi: 10.1016/j.pharmthera.2004.10.012. Pharmacol Ther. 2005. PMID: 15626456 Free PMC article. Review.
-
Parasagittally aligned, mGluR1-dependent patches are evoked at long latencies by parallel fiber stimulation in the mouse cerebellar cortex in vivo.J Neurophysiol. 2011 Apr;105(4):1732-46. doi: 10.1152/jn.00717.2010. Epub 2011 Feb 2. J Neurophysiol. 2011. PMID: 21289138 Free PMC article.
References
-
- Barbour B. Synaptic currents evoked in Purkinje cells by stimulating individual granule cells. Neuron. 1993;11:759–769. - PubMed
-
- Castillo PE, Janz R, Südhof TC, Tzounopoulos T, Malenka RC, Nicoll RA. Rab3A is essential for mossy fibre long-term potentiation in the hippocampus. Nature. 1997;388:590–593. - PubMed
-
- Chavis P, Mollard P, Bockaert J, Manzoni O. Visualization of cyclic AMP-regulated presynaptic activity at cerebellar granule cells. Neuron. 1998;20:773–781. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
