Putative role for interleukin-7 in the maintenance of the recirculating naive CD4+ T-cell pool
- PMID: 10583600
- PMCID: PMC2326939
- DOI: 10.1046/j.1365-2567.1999.00906.x
Putative role for interleukin-7 in the maintenance of the recirculating naive CD4+ T-cell pool
Abstract
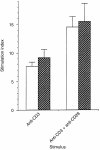
The capacity of the immune system to respond efficiently to new antigens depends upon a continuous source of naive CD4+ T cells. Such cells exit from the thymus and join the recirculated T-cell pool. Factors present at the sites of naive CD4+ T-cell circulation must be responsible for their survival, since upon removal from their host, naive CD4+ T cells die. However, such factors remain unknown. The presence of the cytokine interleukin-7 (IL-7) in secondary lymphoid organs and the continuous expression of its receptor on naive CD4+ T cells prompted us to examine the possibility that IL-7 might be a survival factor for naive CD4+ T cells. Using naive CD4+ T cells isolated from cord blood we show that IL-7, but not IL-2, can maintain naive CD4+ T-cell viability in vitro for at least 15 days. In addition, we find that IL-7 can induce modest proliferation of naive CD4+ T cells without affecting either their cell surface phenotype or their ability to respond to antigenic stimulation. We also find that after anti-CD3 stimulation, naive CD4+ T cells lose that ability to respond to IL-7. However, if cells are primed with IL-7 prior to antigenic stimulation, their proliferative responses are enhanced. Together, these data suggest a novel and important role for IL-7 in the maintenance and maturation of naive CD4+ T cells, ensuring that they can respond maximally when they first meet antigen in secondary lymphoid tissue.
Figures


Similar articles
-
Naive CD4+ T cells can be sensitized with IL-7.Scand J Immunol. 2001 Nov;54(5):501-5. doi: 10.1046/j.1365-3083.2001.01001.x. Scand J Immunol. 2001. PMID: 11696202
-
Interleukin-7 activates human naive CD4+ cells and primes for interleukin-4 production.Eur J Immunol. 1997 Mar;27(3):633-40. doi: 10.1002/eji.1830270309. Eur J Immunol. 1997. PMID: 9079802
-
IL-7/STAT5 cytokine signaling pathway is essential but insufficient for maintenance of naive CD4 T cell survival in peripheral lymphoid organs.J Immunol. 2007 Jan 1;178(1):262-70. doi: 10.4049/jimmunol.178.1.262. J Immunol. 2007. PMID: 17182563
-
Naive and Memory CD4⁺ T Cells Are Differentially Affected in Indonesian HIV Patients Responding to ART.Viral Immunol. 2016 Apr;29(3):176-83. doi: 10.1089/vim.2015.0108. Viral Immunol. 2016. PMID: 27035638
-
IL-7 promotes the survival and maturation but not differentiation of human post-thymic CD4+ T cells.Eur J Immunol. 1998 Oct;28(10):3057-65. doi: 10.1002/(SICI)1521-4141(199810)28:10<3057::AID-IMMU3057>3.0.CO;2-Z. Eur J Immunol. 1998. PMID: 9808174
Cited by
-
IL-7 is critical for homeostatic proliferation and survival of naive T cells.Proc Natl Acad Sci U S A. 2001 Jul 17;98(15):8732-7. doi: 10.1073/pnas.161126098. Epub 2001 Jul 10. Proc Natl Acad Sci U S A. 2001. PMID: 11447288 Free PMC article.
-
Local delivery of interleukin 7 with an oncolytic adenovirus activates tumor-infiltrating lymphocytes and causes tumor regression.Oncoimmunology. 2022 Jul 12;11(1):2096572. doi: 10.1080/2162402X.2022.2096572. eCollection 2022. Oncoimmunology. 2022. PMID: 35845722 Free PMC article.
-
Diminished plasma levels of common γ-chain cytokines in pulmonary tuberculosis and reversal following treatment.PLoS One. 2017 Apr 27;12(4):e0176495. doi: 10.1371/journal.pone.0176495. eCollection 2017. PLoS One. 2017. PMID: 28448542 Free PMC article.
-
Progressive activation of CD127+132- recent thymic emigrants into terminally differentiated CD127-132+ T-cells in HIV-1 infection.PLoS One. 2012;7(2):e31148. doi: 10.1371/journal.pone.0031148. Epub 2012 Feb 13. PLoS One. 2012. PMID: 22348045 Free PMC article.
-
A central role for mTOR kinase in homeostatic proliferation induced CD8+ T cell memory and tumor immunity.Immunity. 2011 Apr 22;34(4):541-53. doi: 10.1016/j.immuni.2011.04.006. Immunity. 2011. PMID: 21511183 Free PMC article.
References
-
- Sprent J, Tough DF. Lymphocyte life‐span and memory. Science. 1994;265:1395. - PubMed
-
- Miller JFAP, Mitchell GF. Thymus and antigen‐reactive cells. Transplantation Rev. 1969;1:3. - PubMed
-
- Cao W, Shores EW, Hu‐Li J, et al. Defective lymphoid development in mice lacking expression of the common cytokine receptor gamma chain. Immunity. 1995;2:223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

