Acinar cell apoptosis and the origin of tubular complexes in caerulein-induced pancreatitis
- PMID: 10583630
- PMCID: PMC2517773
- DOI: 10.1046/j.1365-2613.1999.00116.x
Acinar cell apoptosis and the origin of tubular complexes in caerulein-induced pancreatitis
Abstract
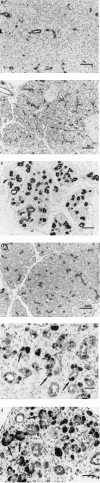
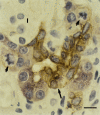
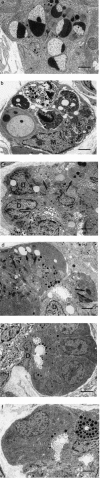
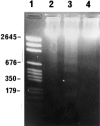
The interrelationship between acinar cell apoptosis and tubular complex formation was examined in caerulein-induced pancreatitis using histology, immunohistochemistry, electron microscopy and DNA gel electrophoresis. Rats were given 8 hourly subcutaneous injections of caerulein, 24 micrograms/kg, for up to 2 days. Morphologically and biochemically typical apoptosis affected 4.6 and 8.9% of acinar cells at 1 and 2 days, respectively, resulting in removal of most acinar cells by 2 days. Consequently, pancreatic ducts, the lining cells expressing bcl-2 and therefore resistant to apoptosis, became much more closely approximated to form the basis of tubular complexes; small numbers of immunohistochemically discrete acinar cells in their lining were either pre-apoptotic resistant to it or newly formed. Proliferation of duct-like lining cells was associated with apoptosis, an increase in islet cells and acinar cell regeneration. There was evidence of duct to acinar cell differentiation but the main increase in acinar cell numbers appeared to derive from proliferation of newly formed acinar cells.
Figures

Similar articles
-
Expression of galectin-3 in the rat pancreas during regeneration following hormone-induced pancreatitis.Cell Tissue Res. 2004 Mar;315(3):321-9. doi: 10.1007/s00441-003-0850-x. Epub 2004 Jan 28. Cell Tissue Res. 2004. PMID: 14747941
-
Temporal relationship of acinar and microvascular changes in caerulein-induced pancreatitis.Br J Surg. 1993 Sep;80(9):1174-6. doi: 10.1002/bjs.1800800936. Br J Surg. 1993. PMID: 8402125
-
Growing pancreatic acinar cells (postpancreatitis and fetal) express a ductal antigen.Pancreas. 1990 Jul;5(4):381-8. doi: 10.1097/00006676-199007000-00002. Pancreas. 1990. PMID: 2199965
-
Morphology of the exocrine pancreas related to pancreatitis.Microsc Res Tech. 1997 Jun 1-15;37(5-6):509-19. doi: 10.1002/(SICI)1097-0029(19970601)37:5/6<509::AID-JEMT13>3.0.CO;2-U. Microsc Res Tech. 1997. PMID: 9220428 Review.
-
Apoptosis of pancreatic acinar cells in acute pancreatitis: is it good or bad?J Cell Mol Med. 2004 Jul-Sep;8(3):402-9. doi: 10.1111/j.1582-4934.2004.tb00330.x. J Cell Mol Med. 2004. PMID: 15491516 Free PMC article. Review.
Cited by
-
PERK eIF2 alpha kinase is required to regulate the viability of the exocrine pancreas in mice.BMC Cell Biol. 2007 Aug 29;8:38. doi: 10.1186/1471-2121-8-38. BMC Cell Biol. 2007. PMID: 17727724 Free PMC article.
-
Luteolin inhibits pancreatitis‑induced acinar‑ductal metaplasia, proliferation and epithelial‑mesenchymal transition of acinar cells.Mol Med Rep. 2018 Mar;17(3):3681-3689. doi: 10.3892/mmr.2017.8327. Epub 2017 Dec 20. Mol Med Rep. 2018. PMID: 29286098 Free PMC article.
-
Cellular expression of Bcl-2 and Bax in atrophic submandibular glands of rats.Int J Exp Pathol. 2008 Oct;89(5):303-8. doi: 10.1111/j.1365-2613.2008.00613.x. Int J Exp Pathol. 2008. PMID: 18808524 Free PMC article.
-
Anterior Gradient 2 (AGR2) Induced Epidermal Growth Factor Receptor (EGFR) Signaling Is Essential for Murine Pancreatitis-Associated Tissue Regeneration.PLoS One. 2016 Oct 20;11(10):e0164968. doi: 10.1371/journal.pone.0164968. eCollection 2016. PLoS One. 2016. PMID: 27764193 Free PMC article.
-
Bone Marrow-Derived Mesenchymal Stem Cells Repair Necrotic Pancreatic Tissue and Promote Angiogenesis by Secreting Cellular Growth Factors Involved in the SDF-1 α /CXCR4 Axis in Rats.Stem Cells Int. 2015;2015:306836. doi: 10.1155/2015/306836. Epub 2015 Feb 25. Stem Cells Int. 2015. PMID: 25810724 Free PMC article.
References
-
- Adler G, Hupp T, Kern HF. Course and spontaneous regression of acute pancreatitis in the rat. Virchows Arch. A. Pathol. Anat. 1979;382:31–47. - PubMed
-
- Ansari B, Coates PJ, Greenstein BD, Hall PA. In situ end-labelling detects DNA strand breaks in apoptosis and other physiological and pathological states. J. Pathol. 1993;170:1–8. - PubMed
-
- Barres BA, Hart IK, Coles HS, et al. Cell death and control of cell survival in the oligodendrocyte lineage. Cell. 1992;70:31–46. - PubMed
-
- Bendayan M. Concentration of amylase along its secretory pathway in the pancreatic acinar cell as revealed by high resolution immunocytochemistry. Histochem. J. 1984;16:85–108. - PubMed
-
- Bockman DE. Morphology of the exocrine pancreas related to pancreatitis. Microsc. Res. Technical. 1997;37:509–519. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

