Sequence requirements for plasmid nuclear import
- PMID: 10585295
- PMCID: PMC4152905
- DOI: 10.1006/excr.1999.4716
Sequence requirements for plasmid nuclear import
Abstract
The nuclear envelope is a major barrier for nuclear uptake of plasmids and represents one of the most significant unsolved problems of nonviral gene delivery. We have previously shown that the nuclear entry of plasmid DNA is sequence-specific, requiring a 366-bp fragment containing the SV40 origin of replication and early promoter. In this report, we show that, although fragments throughout this region can support varying degrees of nuclear import, the 72-bp repeats of the SV40 enhancer facilitate maximal transport. The functions of the promoter and the origin of replication are not needed for nuclear localization of plasmid DNA. In contrast to the import activity of the SV40 enhancer, two other strong promoter and enhancer sequences, the human cytomegalovirus (CMV) immediate-early promoter and the Rous sarcoma virus LTR, were unable to direct nuclear localization of plasmids. The inability of the CMV promoter to mediate plasmid nuclear import was confirmed by measurement of the CMV promoter-driven expression of green fluorescent protein (GFP) in microinjected cells. At times before cell division, as few as 3 to 10 copies per cell of cytoplasmically injected plasmids containing the SV40 enhancer gave significant GFP expression, while no expression was obtained with more than 1000 copies per cell of plasmids lacking the SV40 sequence. However, the levels of expression were the same for both plasmids after cell division in cytoplasmically injected cells and at all times in nuclear injected cells. Thus, the inclusion this SV40 sequence in nonviral vectors may greatly increase their ability to be transported into the nucleus, especially in nondividing cells.
Copyright 1999 Academic Press.
Figures


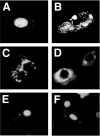
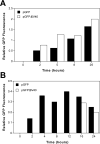
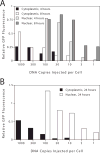

Similar articles
-
Effect of a DNA nuclear targeting sequence on gene transfer and expression of plasmids in the intact vasculature.Gene Ther. 2003 Aug;10(17):1465-70. doi: 10.1038/sj.gt.3302021. Gene Ther. 2003. PMID: 12900761 Free PMC article.
-
Nuclear targeting of plasmid DNA in human corneal cells.Curr Eye Res. 1999 Jul;19(1):66-75. doi: 10.1076/ceyr.19.1.66.5344. Curr Eye Res. 1999. PMID: 10415459
-
Interference of the simian virus 40 origin of replication by the cytomegalovirus immediate early gene enhancer: evidence for competition of active regulatory chromatin conformation in a single domain.Mol Cell Biol. 2000 Jun;20(11):4062-74. doi: 10.1128/MCB.20.11.4062-4074.2000. Mol Cell Biol. 2000. PMID: 10805748 Free PMC article.
-
Nuclear entry of nonviral vectors.Gene Ther. 2005 Jun;12(11):881-90. doi: 10.1038/sj.gt.3302534. Gene Ther. 2005. PMID: 15908994 Free PMC article. Review.
-
Progress and prospects: nuclear import of nonviral vectors.Gene Ther. 2010 Apr;17(4):439-47. doi: 10.1038/gt.2010.31. Epub 2010 Mar 4. Gene Ther. 2010. PMID: 20200566 Free PMC article. Review.
Cited by
-
Non-viral transfection vectors: are hybrid materials the way forward?Medchemcomm. 2019 Aug 16;10(10):1692-1718. doi: 10.1039/c9md00275h. eCollection 2019 Oct 1. Medchemcomm. 2019. PMID: 32180915 Free PMC article. Review.
-
Nanotechnology approaches for gene transfer.Genetica. 2009 Sep;137(1):47-56. doi: 10.1007/s10709-009-9372-0. Epub 2009 Jun 2. Genetica. 2009. PMID: 19488829 Review.
-
Recruitment and rejoining of remote double-strand DNA breaks for enhanced and precise chromosome editing.Genome Biol. 2025 Mar 11;26(1):53. doi: 10.1186/s13059-025-03523-8. Genome Biol. 2025. PMID: 40069752 Free PMC article.
-
Transcription factor plasmid binding modulates microtubule interactions and intracellular trafficking during gene transfer.Gene Ther. 2012 Mar;19(3):338-46. doi: 10.1038/gt.2011.96. Epub 2011 Jun 30. Gene Ther. 2012. PMID: 21716302 Free PMC article.
-
Nonviral DNA vectors for immunization and therapy: design and methods for their obtention.J Mol Med (Berl). 2004 Aug;82(8):500-9. doi: 10.1007/s00109-004-0548-x. Epub 2004 Jun 3. J Mol Med (Berl). 2004. PMID: 15175860 Review.
References
-
- Zabner J, Fasbender AJ, Moninger T, Poellinger KA, Welsh MJ. Cellular and molecular barriers to gene transfer by a cationic lipid. J Biol Chem. 1995;270:18997–19007. - PubMed
-
- Dean DA. Import of plasmid DNA into the nucleus is sequence specific. Exp Cell Res. 1997;230:293–302. - PubMed
-
- Hirt B. Selective extraction of polyoma DNA from infected mouse cell cultures. J Mol Biol. 1967;26:365–369. - PubMed
-
- Tooze J. Molecular biology of tumor viruses, 2nd edition. Cold Spring Harbor Laboratory, Cold Spring Harbor; NY: 1980.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

