Resistance of young gelatinase B-deficient mice to experimental autoimmune encephalomyelitis and necrotizing tail lesions
- PMID: 10587514
- PMCID: PMC409857
- DOI: 10.1172/JCI6886
Resistance of young gelatinase B-deficient mice to experimental autoimmune encephalomyelitis and necrotizing tail lesions
Abstract
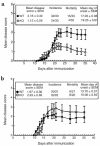
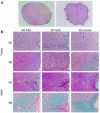
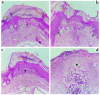
Regulated expression of matrix metalloproteinases (MMPs) and their inhibitors (TIMPs) plays a role in various physiological processes. To determine in vivo how unbalanced expression of these factors can promote or affect the course of pathologies, we knocked out the mouse gelatinase B gene by replacing the catalytic and zinc-binding domains with an antisense-oriented neomycin resistance gene. Adult gelatinase B-deficient mice and wild-type controls could be induced to develop experimental autoimmune encephalomyelitis (EAE) with similar scores for neurologic disease, blood-brain barrier permeability, and central nervous system histopathology. However, whereas diseased control animals showed necrotizing tail lesions with hyperplasia of osteocartilaginous tissue, adult gelatinase B-deficient mice were resistant to this tail pathology. Gelatinase B-deficient mice younger than 4 weeks of age were significantly less susceptible to the development of EAE than were age matched controls and, even as they aged, they remained resistant to tail lesions. These data illustrate that gelatinase B expression plays a role in the development of the immune system and that, in ontogenesis, the propensity to develop autoimmunity is altered by the absence of this MMP.
Figures

Similar articles
-
Gelatinase B/matrix metalloproteinase-9 is a phase-specific effector molecule, independent from Fas, in experimental autoimmune encephalomyelitis.PLoS One. 2018 Oct 1;13(10):e0197944. doi: 10.1371/journal.pone.0197944. eCollection 2018. PLoS One. 2018. PMID: 30273366 Free PMC article.
-
Differential expression of matrix metalloproteinase and tissue inhibitor of matrix metalloproteinase genes in the mouse central nervous system in normal and inflammatory states.Am J Pathol. 1998 Mar;152(3):729-41. Am J Pathol. 1998. PMID: 9502415 Free PMC article.
-
Imaging matrix metalloproteinase activity in multiple sclerosis as a specific marker of leukocyte penetration of the blood-brain barrier.Sci Transl Med. 2016 Nov 9;8(364):364ra152. doi: 10.1126/scitranslmed.aaf8020. Sci Transl Med. 2016. PMID: 27831901
-
Gelatinase B functions as regulator and effector in leukocyte biology.J Leukoc Biol. 2001 Jun;69(6):851-9. J Leukoc Biol. 2001. PMID: 11404367 Review.
-
The gelatinases, MMP-2 and MMP-9, as fine tuners of neuroinflammatory processes.Matrix Biol. 2019 Jan;75-76:102-113. doi: 10.1016/j.matbio.2017.11.007. Epub 2017 Nov 21. Matrix Biol. 2019. PMID: 29158162 Review.
Cited by
-
A further TWEAK to multiple sclerosis pathophysiology.Mol Neurobiol. 2014 Feb;49(1):78-87. doi: 10.1007/s12035-013-8490-y. Epub 2013 Jul 20. Mol Neurobiol. 2014. PMID: 23873135 Review.
-
The ERK1/2 Inhibitor U0126 Attenuates Diabetes-Induced Upregulation of MMP-9 and Biomarkers of Inflammation in the Retina.J Diabetes Res. 2013;2013:658548. doi: 10.1155/2013/658548. Epub 2013 Apr 10. J Diabetes Res. 2013. PMID: 23671886 Free PMC article.
-
Association of matrix metalloproteinase-9 in eosinophilic meningitis of BALB/c mice caused by Angiostrongylus cantonensis.Parasitol Res. 2004 Nov;94(5):321-8. doi: 10.1007/s00436-004-1196-3. Epub 2004 Sep 21. Parasitol Res. 2004. PMID: 15449178
-
Differential gene expression in pristane-induced arthritis susceptible DA versus resistant E3 rats.Arthritis Res Ther. 2003;5(6):R361-72. doi: 10.1186/ar993. Epub 2003 Oct 2. Arthritis Res Ther. 2003. PMID: 14680511 Free PMC article.
-
Matrix metalloproteinases in the CNS: interferons get nervous.Cell Mol Life Sci. 2019 Aug;76(16):3083-3095. doi: 10.1007/s00018-019-03171-9. Epub 2019 Jun 4. Cell Mol Life Sci. 2019. PMID: 31165203 Free PMC article. Review.
References
-
- Agren MS, Jorgensen LN, Andersen M, Viljanto J, Gottrup F. Matrix metalloproteinase 9 level predicts optimal collagen deposition during early wound repair in humans. Br J Surg. 1998;85:68–71. - PubMed
-
- Tsafriri A. Ovulation as a tissue remodelling process. Proteolysis and cumulus expansion. Adv Exp Med Biol. 1995;377:121–140. - PubMed
-
- Sharkey ME, Adler RR, Nieder GL, Brenner CA. Matrix metalloproteinase expression during mouse peri-implantation development. Am J Reprod Immunol. 1996;36:72–80. - PubMed
-
- Vu, T.H., and Werb, Z. 1998. Gelatinase B: structure, regulation, and function. In Matrix metalloproteinases. W.C. Parks and R.P. Mecham, editors. Academic Press. San Diego, CA. 115–137.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

