Cellular compartments of human immunodeficiency virus type 1 replication in vivo: determination by presence of virion-associated host proteins and impact of opportunistic infection
- PMID: 10590100
- PMCID: PMC111522
Cellular compartments of human immunodeficiency virus type 1 replication in vivo: determination by presence of virion-associated host proteins and impact of opportunistic infection
Abstract
Antigens derived from host cells are detectable in the envelope of human immunodeficiency virus type 1 (HIV-1) and result in a distinctive viral phenotype reflecting that of the host cell. An immunomagnetic capture assay targeting discriminatory host proteins was developed to differentiate between HIV-1 derived from macrophages and lymphocytes. HIV-1 propagated in macrophages or lymphocytes in vitro was selectively captured by monoclonal antibodies directed against the virally incorporated cell-type-specific host markers CD36 (macrophages) and CD26 (lymphocytes). Furthermore, by targeting these markers, virus of defined cellular origin was selectively captured from a mixed pool of in vitro-propagated viruses. This technique was further refined in order to determine the impact of opportunistic infection on HIV-1 expression from these cellular compartments in vivo. Analysis of cell-free virus purified from plasma of patients with HIV-1 infection suggested that in those with an opportunistic infection, viral replication occurred in activated lymphocytes. Interestingly, there was also significant replication in activated macrophages in those patients with untreated pulmonary tuberculosis. Thus, in addition to lymphocytes, the macrophage cellular pool may serve as an important source of cell-free HIV-1 in patients with opportunistic infections that lead to marked macrophage activation. This novel viral capture technique may allow researchers to address a wide range of important questions regarding virus-host dynamics.
Figures

 ) was captured selectively by use of anti-CD3, anti-CD25, and anti-CD26. Antibodies to antigens common to both T lymphocytes and macrophages (HLA-A/B/C, HLA-DR, and CD44) captured both viral stocks. Data are representative of three independent experiments with <20% variability in the magnitude of capture.
) was captured selectively by use of anti-CD3, anti-CD25, and anti-CD26. Antibodies to antigens common to both T lymphocytes and macrophages (HLA-A/B/C, HLA-DR, and CD44) captured both viral stocks. Data are representative of three independent experiments with <20% variability in the magnitude of capture.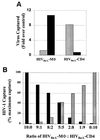
 ) and not by anti-CD36, indicating a discriminating phenotype for identifying the cellular origin of viral replication. (B) HIV-1Ba-L-MΦ and HIV-1Ba-L-CD4 isolates were mixed at various ratios and then captured with both anti-CD36 (■) and anti-CD26 (
) and not by anti-CD36, indicating a discriminating phenotype for identifying the cellular origin of viral replication. (B) HIV-1Ba-L-MΦ and HIV-1Ba-L-CD4 isolates were mixed at various ratios and then captured with both anti-CD36 (■) and anti-CD26 ( ). The amount of virus captured by each antibody was proportional to the input of each type of virus, further illustrating the selective capture of virus derived from diverse cell types. Data are representative of three independent experiments.
). The amount of virus captured by each antibody was proportional to the input of each type of virus, further illustrating the selective capture of virus derived from diverse cell types. Data are representative of three independent experiments.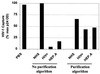
Similar articles
-
Anatomically compartmentalized human immunodeficiency virus replication in HLA-DR+ cells and CD14+ macrophages at the site of pleural tuberculosis coinfection.J Infect Dis. 2001 Nov 1;184(9):1127-33. doi: 10.1086/323649. Epub 2001 Sep 25. J Infect Dis. 2001. PMID: 11598835
-
Co-infection with opportunistic pathogens promotes human immunodeficiency virus type 1 infection in macrophages.J Infect Dis. 1999 May;179 Suppl 3:S457-60. doi: 10.1086/314814. J Infect Dis. 1999. PMID: 10099119 Review.
-
Preliminary in vitro growth cycle and transmission studies of HIV-1 in an autologous primary cell assay of blood-derived macrophages and peripheral blood mononuclear cells.Virology. 1996 Dec 15;226(2):205-16. doi: 10.1006/viro.1996.0648. Virology. 1996. PMID: 8955040
-
Permissive factors for HIV-1 infection of macrophages.J Leukoc Biol. 2000 Sep;68(3):303-10. J Leukoc Biol. 2000. PMID: 10985244 Review.
-
[Molecular pathogenesis in tuberculosis complicated with AIDS].Kekkaku. 2004 Nov;79(11):659-67. Kekkaku. 2004. PMID: 15729891 Review. Japanese.
Cited by
-
Differential incorporation of CD45, CD80 (B7-1), CD86 (B7-2), and major histocompatibility complex class I and II molecules into human immunodeficiency virus type 1 virions and microvesicles: implications for viral pathogenesis and immune regulation.J Virol. 2001 Jul;75(13):6173-82. doi: 10.1128/JVI.75.13.6173-6182.2001. J Virol. 2001. PMID: 11390619 Free PMC article.
-
Exogenous and endogenous hyaluronic acid reduces HIV infection of CD4(+) T cells.Immunol Cell Biol. 2014 Oct;92(9):770-80. doi: 10.1038/icb.2014.50. Epub 2014 Jun 24. Immunol Cell Biol. 2014. PMID: 24957217 Free PMC article.
-
Nanoparticle-based flow virometry for the analysis of individual virions.J Clin Invest. 2013 Sep;123(9):3716-27. doi: 10.1172/JCI67042. Epub 2013 Aug 8. J Clin Invest. 2013. PMID: 23925291 Free PMC article.
-
Impairment of beta chemokine and cytokine production in patients with HIV related Pneumocystis jerovici pneumonia.Thorax. 2004 Mar;59(3):247-51. doi: 10.1136/thx.2003.013763. Thorax. 2004. PMID: 14985564 Free PMC article.
-
Vpx complementation of 'non-macrophage tropic' R5 viruses reveals robust entry of infectious HIV-1 cores into macrophages.Retrovirology. 2014 Mar 21;11:25. doi: 10.1186/1742-4690-11-25. Retrovirology. 2014. PMID: 24656066 Free PMC article.
References
-
- Arthur L O, Bess J W, Jr, Sowder R C, Benveniste R E, Mann D L, Chermann J C, Henderson L E. Cellular proteins bound to immunodeficiency viruses: implication for pathogenesis and vaccines. Science. 1992;258:1935–1938. - PubMed
-
- Bourinbaiar A S. The ratio of defective HIV-1 particles to replication-competent infectious virions. Acta Virol. 1994;38:59–61. - PubMed
-
- Cantin R, Fortin J-F, Lamontagne G, Tremblay M. The acquisition of host-derived major histocompatibility complex class II glycoproteins by human immunodeficiency virus type 1 accelerates the process of virus entry and infection in human T-lymphoid cells. Blood. 1997;90:1091–1100. - PubMed
-
- Dierich M P, Frank I, Stoiber A, Clivio M, Spruth M, Katinger H W. The envelope of HIV. Immunol Lett. 1996;54:205–206. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
