Gap junctional coupling and patterns of connexin expression among neonatal rat lumbar spinal motor neurons
- PMID: 10594064
- PMCID: PMC6784933
- DOI: 10.1523/JNEUROSCI.19-24-10813.1999
Gap junctional coupling and patterns of connexin expression among neonatal rat lumbar spinal motor neurons
Abstract
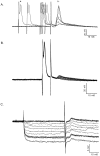
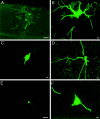
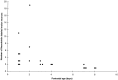
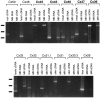
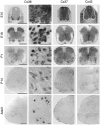
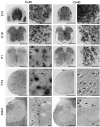
Interneuronal gap junctional coupling is a hallmark of neural development whose functional significance is poorly understood. We have characterized the extent of electrical coupling and dye coupling and patterns of gap junction protein expression in lumbar spinal motor neurons of neonatal rats. Intracellular recordings showed that neonatal motor neurons are transiently electrically coupled and that electrical coupling is reversibly abolished by halothane, a gap junction blocker. Iontophoretic injection of Neurobiotin, a low molecular weight compound that passes across most gap junctions, into single motor neurons resulted in clusters of many labeled motor neurons at postnatal day 0 (P0)-P2, and single labeled motor neurons after P7. The compact distribution of dye-labeled motor neurons suggested that, after birth, gap junctional coupling is spatially restricted. RT-PCR, in situ hybridization, and immunostaining showed that motor neurons express five connexins, Cx36, Cx37, Cx40, Cx43, and Cx45, a repertoire distinct from that expressed by other neurons or glia. Although all five connexins are widely expressed among motor neurons in embryonic and neonatal life, Cx36, Cx37, and Cx43 continue to be expressed in many adult motor neurons, and expression of Cx45, and in particular Cx40, decreases after birth. The disappearance of electrical and dye coupling despite the persistent expression of several gap junction proteins suggests that gap junctional communication among motor neurons may be modulated by mechanisms that affect gap junction assembly, permeability, or open state.
Figures



References
-
- Balice-Gordon RJ, Lichtman JW. Long-term synapse loss induced by focal blockade of postsynaptic receptors. Nature. 1994;372:519–524. - PubMed
-
- Bergoffen J, Scherer SS, Wang S, Scott M, Bone LJ, Paul DL, Chen K, Lensch MW, Chance PF, Fischbeck KH. Connexin mutations in X-linked charcot-marie-tooth disease. Science. 1993;262:2039–2042. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
