Proprioceptive control of extensor activity during fictive scratching and weight support compared to fictive locomotion
- PMID: 10594077
- PMCID: PMC6784935
- DOI: 10.1523/JNEUROSCI.19-24-10966.1999
Proprioceptive control of extensor activity during fictive scratching and weight support compared to fictive locomotion
Abstract
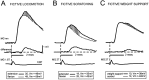
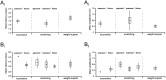
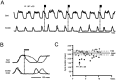
At rest, extensor group I afferents produce oligosynaptic inhibition of extensor motoneurons. During locomotor activity, however, such inhibition is replaced by oligosynaptic excitation. Oligosynaptic excitation from extensor group I afferents plays a crucial role in the regulation of extensor activity during walking. In this study we investigate the possibility that this mechanism also regulates extensor muscle activity during other motor tasks. We show that the reflex pathways responsible for extensor group I oligosynaptic excitation during fictive locomotion can be activated during both fictive scratching and fictive weight support (tonic motor activity induced by contralateral scratching). These observations suggest that the excitatory group I oligosynaptic reflex pathways are open for transmission during several forms of motor activities. We also show that extensor group I input during fictive scratching can affect the amplitude and the timing of extensor activity in a pattern similar to that observed during locomotion. Most likely these effects involve the activation of the excitatory group I oligosynaptic reflex pathways. Accordingly, it is suggested that extensor group I oligosynaptic excitation during motor activities other than locomotion is also used to regulate extensor muscle activity. Furthermore, the similarity of effects from extensor group I input on the rhythmicity during scratching and locomotion supports the hypothesis that both rhythms are generated by a common network.
Figures




Similar articles
-
Modulation of oligosynaptic cutaneous and muscle afferent reflex pathways during fictive locomotion and scratching in the cat.J Neurophysiol. 1998 Jan;79(1):447-63. doi: 10.1152/jn.1998.79.1.447. J Neurophysiol. 1998. PMID: 9425213
-
Differential modulation of disynaptic cutaneous inhibition and excitation in ankle flexor motoneurons during fictive locomotion.J Neurophysiol. 1996 Nov;76(5):2972-85. doi: 10.1152/jn.1996.76.5.2972. J Neurophysiol. 1996. PMID: 8930248
-
Modulation of short latency cutaneous excitation in flexor and extensor motoneurons during fictive locomotion in the cat.Exp Brain Res. 1989;77(1):57-68. doi: 10.1007/BF00250567. Exp Brain Res. 1989. PMID: 2792270
-
Neuronal basis of afferent-evoked enhancement of locomotor activity.Ann N Y Acad Sci. 1998 Nov 16;860:216-25. doi: 10.1111/j.1749-6632.1998.tb09051.x. Ann N Y Acad Sci. 1998. PMID: 9928314 Review.
-
Proprioceptive regulation of locomotion.Curr Opin Neurobiol. 1995 Dec;5(6):786-91. doi: 10.1016/0959-4388(95)80107-3. Curr Opin Neurobiol. 1995. PMID: 8805415 Review.
Cited by
-
Modeling Post-Scratching Locomotion with Two Rhythm Generators and a Shared Pattern Formation.Biology (Basel). 2021 Jul 14;10(7):663. doi: 10.3390/biology10070663. Biology (Basel). 2021. PMID: 34356518 Free PMC article.
-
Motoneurons have different membrane resistance during fictive scratching and weight support.J Neurosci. 2002 Sep 15;22(18):8259-65. doi: 10.1523/JNEUROSCI.22-18-08259.2002. J Neurosci. 2002. PMID: 12223580 Free PMC article.
-
Reciprocal Ia inhibition contributes to motoneuronal hyperpolarisation during the inactive phase of locomotion and scratching in the cat.J Physiol. 2011 Jan 1;589(Pt 1):119-34. doi: 10.1113/jphysiol.2010.199125. Epub 2010 Nov 8. J Physiol. 2011. PMID: 21059756 Free PMC article.
-
Afferent control of locomotor CPG: insights from a simple neuromechanical model.Ann N Y Acad Sci. 2010 Jun;1198:21-34. doi: 10.1111/j.1749-6632.2010.05435.x. Ann N Y Acad Sci. 2010. PMID: 20536917 Free PMC article.
-
Information to cerebellum on spinal motor networks mediated by the dorsal spinocerebellar tract.J Physiol. 2013 Nov 15;591(22):5433-43. doi: 10.1113/jphysiol.2012.249110. Epub 2013 Apr 22. J Physiol. 2013. PMID: 23613538 Free PMC article. Review.
References
-
- Baev KV, Kostyuk PG. Primary afferent depolarisation evoked by the activity of spinal scratching generator. Neuroscience. 1981;6:205–215. - PubMed
-
- Baev KV, Esipenko VB, Shimansky YP. Afferent control of central pattern generators: experimental analysis of scratching in the decerebrate cat. Neuroscience. 1991;40:239–256. - PubMed
-
- Berkinblit MB, Deliagina TG, Orlovsky GN, Feldman AG. Activity of motoneurons during fictitious scratch reflex in the cat. Brain Res. 1980;193:427–438. - PubMed
-
- Brownstone RM, Gossard JP, Hultborn H. Voltage-dependent excitation of motoneurones from spinal locomotor centres in the cat. Exp Brain Res. 1994;102:34–44. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
