Synaptophysin: A novel marker for human and rat hepatic stellate cells
- PMID: 10595912
- PMCID: PMC1866940
- DOI: 10.1016/S0002-9440(10)65501-0
Synaptophysin: A novel marker for human and rat hepatic stellate cells
Abstract
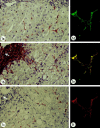
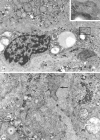
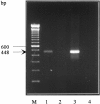
Synaptophysin is a protein involved in neurotransmitter exocytosis and is a neuroendocrine marker. We studied synaptophysin immunohistochemical expression in 35 human liver specimens (normal and different pathological conditions), in rat models of galactosamine hepatitis and carbon tetrachloride-induced cirrhosis, and in freshly isolated rat stellate cells. Synaptophysin reactivity was present in perisinusoidal stellate cells in both human and rat normal liver biopsies. The number of synaptophysin-reactive perisinusoidal cells increased in pathological conditions. Double staining for alpha-smooth muscle actin and synaptophysin, detected by confocal laser scanning microscopy, unequivocally demonstrated colocalization of both markers in lobular stellate cells. In addition, freshly isolated rat stellate cells expressed synaptophysin mRNA (detected by polymerase chain reaction) and protein. Finally, electron microscopy showed the presence of small electron translucent vesicles, comparable to the synaptophysin-reactive synaptic vesicles in neurons, in stellate cell projections. We conclude that synaptophysin is a novel marker for quiescent as well as activated hepatic stellate cells. Together with the stellate cell's expression of neural cell adhesion molecule, glial fibrillary acidic protein, and nestin, this finding raises questions about its embryonic origin and its differentiation. In addition, the presence of synaptic vesicles in stellate cell processes suggests a hitherto unknown mechanism of interaction with neighboring cells.
Figures

Similar articles
-
Class VI intermediate filament protein nestin is induced during activation of rat hepatic stellate cells.Hepatology. 1999 Feb;29(2):520-7. doi: 10.1002/hep.510290232. Hepatology. 1999. PMID: 9918930
-
Human and rat hepatic stellate cells express neurotrophins and neurotrophin receptors.Hepatology. 2001 Jan;33(1):148-58. doi: 10.1053/jhep.2001.20793. Hepatology. 2001. PMID: 11124831
-
GFAP expression in the liver as an early marker of stellate cells activation.Ital J Anat Embryol. 2005 Oct-Dec;110(4):193-207. Ital J Anat Embryol. 2005. PMID: 16536051
-
Neuroregulation of the neuroendocrine compartment of the liver.Anat Rec A Discov Mol Cell Evol Biol. 2004 Sep;280(1):910-23. doi: 10.1002/ar.a.20096. Anat Rec A Discov Mol Cell Evol Biol. 2004. PMID: 15382010 Review.
-
[A synaptic marker synaptophysin].Morfologiia. 2015;147(1):78-82. Morfologiia. 2015. PMID: 25958734 Review. Russian.
Cited by
-
Targeting liver myofibroblasts: a novel approach in anti-fibrogenic therapy.Hepatol Int. 2008 Dec;2(4):405-15. doi: 10.1007/s12072-008-9093-y. Epub 2008 Sep 3. Hepatol Int. 2008. PMID: 19669316 Free PMC article.
-
Morphological characterisation of portal myofibroblasts and hepatic stellate cells in the normal dog liver.Comp Hepatol. 2006 Nov 16;5:7. doi: 10.1186/1476-5926-5-7. Comp Hepatol. 2006. PMID: 17109742 Free PMC article.
-
Hepatic Innervations and Nonalcoholic Fatty Liver Disease.Semin Liver Dis. 2023 May;43(2):149-162. doi: 10.1055/s-0043-57237. Epub 2023 May 8. Semin Liver Dis. 2023. PMID: 37156523 Free PMC article. Review.
-
Hepatic stellate cells - from past till present: morphology, human markers, human cell lines, behavior in normal and liver pathology.Rom J Morphol Embryol. 2020 Jul-Sep;61(3):615-642. doi: 10.47162/RJME.61.3.01. Rom J Morphol Embryol. 2020. PMID: 33817704 Free PMC article. Review.
-
A new fluorescence-based optical imaging method to non-invasively monitor hepatic myofibroblasts in vivo.J Hepatol. 2016 Jul;65(1):75-83. doi: 10.1016/j.jhep.2016.03.021. Epub 2016 Apr 9. J Hepatol. 2016. PMID: 27067455 Free PMC article.
References
-
- Jonker AM, Dijkhuis FW, Kroese FG, Hardonk MJ, Grond J: Immunopathology of acute galactosamine hepatitis in rats. Hepatology 1990, 11:622-627 - PubMed
-
- Johnson SJ, Hines JE, Burt AD: Macrophage and perisinusoidal cell kinetics in acute liver injury. J Pathol 1992, 166:351-358 - PubMed
-
- Yokoi Y, Namihisa T, Matsuzaki K, Miyazaki A, Yamaguchi Y: Distribution of Ito cells in experimental hepatic fibrosis. Liver 1988, 8:48-52 - PubMed
-
- Geerts A, Lazou JM, De Bleser P, Wisse E: Tissue distribution, quantitation and proliferation kinetics of fat-storing cells in carbon tetrachloride-injured rat liver. Hepatology 1991, 13:1193-1202 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

