A transgenic rabbit model for human hypertrophic cardiomyopathy
- PMID: 10606622
- PMCID: PMC409884
- DOI: 10.1172/JCI7956
A transgenic rabbit model for human hypertrophic cardiomyopathy
Abstract
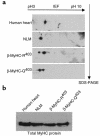
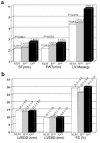
Certain mutations in genes for sarcomeric proteins cause hypertrophic cardiomyopathy (HCM). We have developed a transgenic rabbit model for HCM caused by a common point mutation in the beta-myosin heavy chain (MyHC) gene, R400Q. Wild-type and mutant human beta-MyHC cDNAs were cloned 3' to a 7-kb murine beta-MyHC promoter. We injected purified transgenes into fertilized zygotes to generate two lines each of the wild-type and mutant transgenic rabbits. Expression of transgene mRNA and protein were confirmed by Northern blotting and 2-dimensional gel electrophoresis followed by immunoblotting, respectively. Animals carrying the mutant transgene showed substantial myocyte disarray and a 3-fold increase in interstitial collagen expression in their myocardia. Mean septal thicknesses were comparable between rabbits carrying the wild type transgene and their nontransgenic littermates (NLMs) but were significantly increased in the mutant transgenic animals. Posterior wall thickness and left ventricular mass were also increased, but dimensions and systolic function were normal. Premature death was more common in mutant than in wild-type transgenic rabbits or in NLMs. Thus, cardiac expression of beta-MyHC-Q(403) in transgenic rabbits induced hypertrophy, myocyte and myofibrillar disarray, interstitial fibrosis, and premature death, phenotypes observed in humans patients with HCM due to beta-MyHC-Q(403).
Figures




References
-
- Maron BJ, et al. Prevalence of hypertrophic cardiomyopathy in a general population of young adults. Echocardiographic analysis of 4111 subjects in the CARDIA Study. Coronary Artery Risk Development in (Young) Adults. Circulation. 1995;92:785–789. - PubMed
-
- Wigle ED, Rakowski H, Kimball BP, Williams WG. Hypertrophic cardiomyopathy. Clinical spectrum and treatment. Circulation. 1995;92:1680–1692. - PubMed
-
- Ferrans VJ, Rodriguez ER. Specificity of light and electron microscopic features of hypertrophic obstructive and nonobstructive cardiomyopathy. Qualitative, quantitative and etiologic aspects. Eur Heart J. 1983;4(Suppl. F):9–22. - PubMed
-
- Maron BJ, et al. Sudden death in young competitive athletes. Clinical, demographic, and pathological profiles. JAMA. 1996;276:199–204. - PubMed
-
- Marian AJ, Roberts R. Molecular genetic basis of hypertrophic cardiomyopathy: genetic markers for sudden cardiac death. J Cardiovasc Electrophysiol. 1998;9:88–99. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

