Mycobacterial infection of macrophages results in membrane-permeable phagosomes
- PMID: 10611360
- PMCID: PMC24795
- DOI: 10.1073/pnas.96.26.15190
Mycobacterial infection of macrophages results in membrane-permeable phagosomes
Abstract
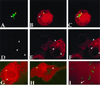
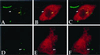
Cell-mediated immunity is critical for host resistance to tuberculosis. T lymphocytes recognizing antigens presented by the major histocompatibility complex (MHC) class I and class II molecules have been found to be necessary for control of mycobacterial infection. Mice genetically deficient in the generation of MHC class I and class Ia responses are susceptible to mycobacterial infection. Although soluble protein antigens are generally presented by macrophages to T cells through MHC class II molecules, macrophages infected with Mycobacterium tuberculosis or bacille Calmette-Guerin have been shown to facilitate presentation of ovalbumin through the MHC class I presentation pathway via a TAP-dependent mechanism. How mycobacteria, thought to reside within membrane-bound vacuoles, facilitate communication with the cytoplasm and enable MHC class I presentation presents a paradox. By using confocal microscopy to study the localization of fluorescent-tagged dextrans of varying size microinjected intracytoplasmically into macrophages infected with bacille Calmette-Guerin expressing the green fluorescent protein, molecules as large as 70 kilodaltons were shown to gain access to the mycobacterial phagosome. Possible biological consequences of the permeabilization of vacuolar membranes by mycobacteria would be pathogen access to host cell nutrients within the cytoplasm, perhaps contributing to bacterial pathogenesis, and access of microbial antigens to the MHC class I presentation pathway, contributing to host protective immune responses.
Figures

Similar articles
-
PPE38 Protein of Mycobacterium tuberculosis Inhibits Macrophage MHC Class I Expression and Dampens CD8+ T Cell Responses.Front Cell Infect Microbiol. 2017 Mar 13;7:68. doi: 10.3389/fcimb.2017.00068. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28348981 Free PMC article.
-
Lipoprotein access to MHC class I presentation during infection of murine macrophages with live mycobacteria.J Immunol. 2001 Jan 1;166(1):447-57. doi: 10.4049/jimmunol.166.1.447. J Immunol. 2001. PMID: 11123323
-
Major histocompatibility class I presentation of soluble antigen facilitated by Mycobacterium tuberculosis infection.Proc Natl Acad Sci U S A. 1996 Oct 15;93(21):11786-91. doi: 10.1073/pnas.93.21.11786. Proc Natl Acad Sci U S A. 1996. PMID: 8876215 Free PMC article.
-
Cytotoxic T lymphocytes in resistance to tuberculosis.Adv Exp Med Biol. 1998;452:85-101. doi: 10.1007/978-1-4615-5355-7_11. Adv Exp Med Biol. 1998. PMID: 9889963 Review.
-
Impact of intracellular location of and antigen display by intracellular bacteria: implications for vaccine development.Immunol Lett. 1999 Jan;65(1-2):81-4. doi: 10.1016/s0165-2478(98)00128-x. Immunol Lett. 1999. PMID: 10065631 Review.
Cited by
-
Disruption of the gene homologous to mammalian Nramp1 in Mycobacterium tuberculosis does not affect virulence in mice.Infect Immun. 2002 Aug;70(8):4124-31. doi: 10.1128/IAI.70.8.4124-4131.2002. Infect Immun. 2002. PMID: 12117920 Free PMC article.
-
PPE38 Protein of Mycobacterium tuberculosis Inhibits Macrophage MHC Class I Expression and Dampens CD8+ T Cell Responses.Front Cell Infect Microbiol. 2017 Mar 13;7:68. doi: 10.3389/fcimb.2017.00068. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28348981 Free PMC article.
-
Role of MHC class I pathways in Mycobacterium tuberculosis antigen presentation.Front Cell Infect Microbiol. 2023 Mar 15;13:1107884. doi: 10.3389/fcimb.2023.1107884. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37009503 Free PMC article. Review.
-
Mycobacterial PPE36 Modulates Host Inflammation by Promoting E3 Ligase Smurf1-Mediated MyD88 Degradation.Front Immunol. 2022 Feb 14;13:690667. doi: 10.3389/fimmu.2022.690667. eCollection 2022. Front Immunol. 2022. PMID: 35237255 Free PMC article.
-
The fibrinolytic system in dissemination and matrix protein deposition during a mycobacterium infection.Am J Pathol. 2003 Aug;163(2):517-31. doi: 10.1016/S0002-9440(10)63680-2. Am J Pathol. 2003. PMID: 12875972 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

