Active Fe-containing superoxide dismutase and abundant sodF mRNA in Nostoc commune (Cyanobacteria) after years of desiccation
- PMID: 10613879
- PMCID: PMC94256
- DOI: 10.1128/JB.182.1.189-197.2000
Active Fe-containing superoxide dismutase and abundant sodF mRNA in Nostoc commune (Cyanobacteria) after years of desiccation
Abstract
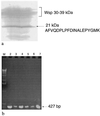
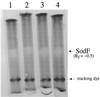
Active Fe-superoxide dismutase (SodF) was the third most abundant soluble protein in cells of Nostoc commune CHEN/1986 after prolonged (13 years) storage in the desiccated state. Upon rehydration, Fe-containing superoxide disumutase (Fe-SOD) was released and the activity was distributed between rehydrating cells and the extracellular fluid. The 21-kDa Fe-SOD polypeptide was purified, the N terminus was sequenced, and the data were used to isolate sodF from the clonal isolate N. commune DRH1. sodF encodes an open reading frame of 200 codons and is expressed as a monocistronic transcript (of approximately 750 bases) from a region of the genome which includes genes involved in nucleic acid synthesis and repair, including dipyrimidine photolyase (phr) and cytidylate monophosphate kinase (panC). sodF mRNA was abundant and stable in cells after long-term desiccation. Upon rehydration of desiccated cells, there was a turnover of sodF mRNA within 15 min and then a rise in the mRNA pool to control levels (quantity of sodF mRNA in cells in late logarithmic phase of growth) over approximately 24 h. The extensive extracellular polysaccharide (glycan) of N. commune DRH1 generated superoxide radicals upon exposure to UV-A or -B irradiation, and these were scavenged by SOD. Despite demonstrated roles for the glycan in the desiccation tolerance of N. commune, it may in fact be a significant source of damaging free radicals in vivo. It is proposed that the high levels of SodF in N. commune, and release of the enzyme from dried cells upon rehydration, counter the effects of oxidative stress imposed by multiple cycles of desiccation and rehydration during UV-A or -B irradiation in situ.
Figures



Similar articles
-
UV irradiation and desiccation modulate the three-dimensional extracellular matrix of Nostoc commune (Cyanobacteria).J Biol Chem. 2005 Dec 2;280(48):40271-81. doi: 10.1074/jbc.M505961200. Epub 2005 Sep 27. J Biol Chem. 2005. PMID: 16192267
-
Characterization of extracellular matrix components from the desiccation-tolerant cyanobacterium Nostoc commune.J Gen Appl Microbiol. 2018 Mar 27;64(1):15-25. doi: 10.2323/jgam.2017.03.001. Epub 2017 Dec 8. J Gen Appl Microbiol. 2018. PMID: 29225284
-
Expression and regulation of the sodF gene encoding iron- and zinc-containing superoxide dismutase in Streptomyces coelicolor Müller.J Bacteriol. 1998 Apr;180(8):2014-20. doi: 10.1128/JB.180.8.2014-2020.1998. J Bacteriol. 1998. PMID: 9555880 Free PMC article.
-
Desiccation tolerance of prokaryotes.Microbiol Rev. 1994 Dec;58(4):755-805. doi: 10.1128/mr.58.4.755-805.1994. Microbiol Rev. 1994. PMID: 7854254 Free PMC article. Review.
-
Healthy efficacy of Nostoc commune Vaucher.Oncotarget. 2017 Dec 22;9(18):14669-14679. doi: 10.18632/oncotarget.23620. eCollection 2018 Mar 6. Oncotarget. 2017. PMID: 29581873 Free PMC article. Review.
Cited by
-
Purification and biochemical characterization of a superoxide dismutase from the soluble fraction of the cyanobacterium, Spirulina platensis.World J Microbiol Biotechnol. 2007 Dec;23(12):1661-6. doi: 10.1007/s11274-007-9413-8. Epub 2007 May 13. World J Microbiol Biotechnol. 2007. PMID: 27517820
-
Immunomodulatory Potential of the Polysaccharide-Rich Extract from Edible Cyanobacterium Nostoc commune.Med Sci (Basel). 2015 Nov 4;3(4):112-123. doi: 10.3390/medsci3040112. Med Sci (Basel). 2015. PMID: 29083396 Free PMC article.
-
Mutations stabilize small subunit ribosomal RNA in desiccation-tolerant cyanobacteria nostoc.Curr Microbiol. 2007 Apr;54(4):254-9. doi: 10.1007/s00284-006-0095-5. Epub 2007 Mar 2. Curr Microbiol. 2007. PMID: 17334839
-
Unusual water flux in the extracellular polysaccharide of the cyanobacterium Nostoc commune.Appl Environ Microbiol. 2003 Sep;69(9):5679-84. doi: 10.1128/AEM.69.9.5679-5684.2003. Appl Environ Microbiol. 2003. PMID: 12957961 Free PMC article.
-
The inhibitory effect of calcium on Cylindrospermopsis raciborskii (cyanobacteria) metabolism.Braz J Microbiol. 2011 Oct;42(4):1547-59. doi: 10.1590/S1517-838220110004000042. Epub 2011 Dec 1. Braz J Microbiol. 2011. PMID: 24031789 Free PMC article.
References
-
- Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K. Short protocols in molecular biology. 2nd ed. New York, N.Y: John Wiley and Sons; 1992. p. I-index 25.
-
- Barnes A C, Balebona M C, Horne M T, Ellis A E. Superoxide dismutase and catalase in Photobacterium damselae subsp. piscicida and their roles in resistance to reactive oxygen species. Microbiology. 1999;145:483–494. - PubMed
-
- Beaman B L, Scates S M, Moring S E, Deem R, Misra H P. Purification and properties of a unique superoxide dismutase from Nocardia asteroides. J Biol Chem. 1983;258:91–96. - PubMed
-
- Beckman K B, Ames B N. The free radical theory of aging matures. Physiol Rev. 1998;78:547–581. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources

