Alkaline phosphatase knock-out mice recapitulate the metabolic and skeletal defects of infantile hypophosphatasia
- PMID: 10620060
- PMCID: PMC3049802
- DOI: 10.1359/jbmr.1999.14.12.2015
Alkaline phosphatase knock-out mice recapitulate the metabolic and skeletal defects of infantile hypophosphatasia
Abstract
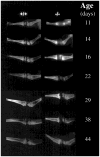
Hypophosphatasia is an inborn error of metabolism characterized by deficient activity of the tissue-nonspecific isoenzyme of alkaline phosphatase (TNSALP) and skeletal disease due to impaired mineralization of cartilage and bone matrix. We investigated two independently generated TNSALP gene knock-out mouse strains as potential models for hypophosphatasia. Homozygous mice (-/-) had < 1% of wild-type plasma TNSALP activity; heterozygotes had the predicted mean of approximately 50%. Phosphoethanolamine, inorganic pyrophosphate, and pyridoxal 5'-phosphate are putative natural substrates for TNSALP and all were increased endogenously in the knock-out mice. Skeletal disease first appeared radiographically at approximately 10 days of age and featured worsening rachitic changes, osteopenia, and fracture. Histologic studies revealed developmental arrest of chondrocyte differentiation in epiphyses and in growth plates with diminished or absent hypertrophic zones. Progressive osteoidosis from defective skeletal matrix mineralization was noted but not associated with features of secondary hyperparathyroidism. Plasma and urine calcium and phosphate levels were unremarkable. Our findings demonstrate that TNSALP knock-out mice are a good model for the infantile form of hypophosphatasia and provide compelling evidence for an important role for TNSALP in postnatal development and mineralization of the murine skeleton.
Figures






References
-
- Whyte MP. Hypophosphatasia. In: Scriver CR, Beaudet AL, Sly WS, Valle D, editors. The Metabolic and Molecular Bases of Inherited Disease. 8. McGraw-Hill; New York, NY, U.S.A: in press.
-
- Whyte MP. Hypophosphatasia and the role of alkaline phosphatase in skeletal mineralization. Endocr Rev. 1994;15:439–461. - PubMed
-
- Zurutuza L, Muller F, Gibrat JF, Traillandier A, Simon-Bouy B, Serre JL, Mornet E. Correlations of genotype and phenotype in hypophosphatasia. Hum Mol Genet. 1999;8:1039–1046. - PubMed
-
- Whyte MP. Hypophosphatasia. In: Econs MC, editor. The Genetics of Osteoporosis and Metabolic Bone Disease. Humana Press, Inc; Totowa, NJ, U.S.A: in press.
-
- Fraser D. Hypophosphatasia. Am J Med. 1957;22:730–746. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

