Stimulation of border cell production in response to increased carbon dioxide levels
- PMID: 10631261
- PMCID: PMC58856
- DOI: 10.1104/pp.122.1.181
Stimulation of border cell production in response to increased carbon dioxide levels
Abstract
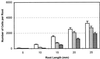
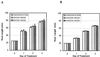
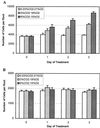
Field soil atmospheres have higher CO(2) and lower O(2) concentrations compared with ambient atmosphere, but little is known about the impact of such conditions on root exudation patterns. We used altered levels of CO(2) and O(2) relative to ambient conditions to examine the influence of the atmosphere on the production of root border cells by pea (Pisum sativum) root tips. During germination, atmospheres with high CO(2) and low O(2) inhibited root development and border cell separation in pea seedlings. Later in development, the same atmospheric composition stimulated border cell separation without significantly influencing root growth. Increased CO(2), not low O(2), was responsible for the observed stimulation of border cell number. High CO(2) apparently can override endogenous signals that regulate the number of border cells released from pea roots into the rhizosphere. The same conditions that stimulated border cell production in pea had no such effect in alfalfa (Medicago sativa).
Figures

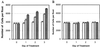
Similar articles
-
Induction of microbial genes for pathogenesis and symbiosis by chemicals from root border cells.Plant Physiol. 1997 Dec;115(4):1691-8. doi: 10.1104/pp.115.4.1691. Plant Physiol. 1997. PMID: 9414568 Free PMC article.
-
Visualization of extracellular DNA released during border cell separation from the root cap.Am J Bot. 2017 Jul;104(7):970-978. doi: 10.3732/ajb.1700142. Epub 2017 Jul 14. Am J Bot. 2017. PMID: 28710125
-
Apoptosis-like programmed cell death occurs in procambium and ground meristem of pea (Pisum sativum) root tips exposed to sudden flooding.Ann Bot. 2006 May;97(5):895-902. doi: 10.1093/aob/mcl040. Epub 2006 Mar 13. Ann Bot. 2006. PMID: 16533830 Free PMC article.
-
Xyloglucan and cellulose form molecular cross-bridges connecting root border cells in pea (Pisum sativum).Plant Physiol Biochem. 2019 Jun;139:191-196. doi: 10.1016/j.plaphy.2019.03.023. Epub 2019 Mar 17. Plant Physiol Biochem. 2019. PMID: 30904720
-
Root border cells as tools in plant cell studies.Methods Cell Biol. 1995;49:377-87. doi: 10.1016/s0091-679x(08)61467-3. Methods Cell Biol. 1995. PMID: 8531770 Review. No abstract available.
Cited by
-
Immobilization of aluminum with mucilage secreted by root cap and root border cells is related to aluminum resistance in Glycine max L.Environ Sci Pollut Res Int. 2013 Dec;20(12):8924-33. doi: 10.1007/s11356-013-1815-6. Epub 2013 Jun 8. Environ Sci Pollut Res Int. 2013. PMID: 23749363
-
Root cap structure and cell production rates of maize (Zea mays) roots in compacted sand.New Phytol. 2003 Oct;160(1):127-134. doi: 10.1046/j.1469-8137.2003.00860.x. New Phytol. 2003. PMID: 33873521
-
Plant Immune Mechanisms: From Reductionistic to Holistic Points of View.Mol Plant. 2020 Oct 5;13(10):1358-1378. doi: 10.1016/j.molp.2020.09.007. Epub 2020 Sep 8. Mol Plant. 2020. PMID: 32916334 Free PMC article. Review.
-
Combinatorial impacts of elevated CO2 and temperature affect growth, development, and fruit yield in Capsicum chinense Jacq.Physiol Mol Biol Plants. 2023 Mar;29(3):393-407. doi: 10.1007/s12298-023-01294-9. Epub 2023 Mar 16. Physiol Mol Biol Plants. 2023. PMID: 37033763 Free PMC article.
-
Salt stress of two rice varieties: root border cell response and multi-logistic quantification.Protoplasma. 2021 Sep;258(5):1119-1131. doi: 10.1007/s00709-021-01629-x. Epub 2021 Mar 6. Protoplasma. 2021. PMID: 33677735
References
-
- Abrosimova LN, Revut IB. Biological activity and the composition of soil air in the plow layer. Sov Soil Sci. 1964;7:682–691.
-
- Bown AW. CO2 and intracellular pH. Plant Cell Environ. 1985;8:459–465.
-
- Buyanovsky GA, Wagner GH. Annual cycles of carbon dioxide level in soil air. Soil Sci Soc Am J. 1983;47:1139–1145.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

