Behavioral evidence of depolarization block of dopamine neurons after chronic treatment with haloperidol and clozapine
- PMID: 10648727
- PMCID: PMC6774188
- DOI: 10.1523/JNEUROSCI.20-03-01229.2000
Behavioral evidence of depolarization block of dopamine neurons after chronic treatment with haloperidol and clozapine
Abstract
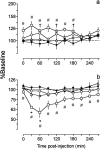
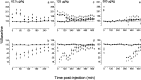
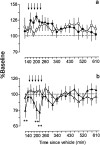
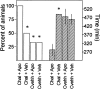
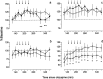
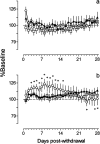
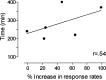
Electrophysiological studies have shown that chronic treatment with haloperidol causes depolarization block (DB) of dopamine cells in anesthetized and paralyzed rats. It has been proposed that the emergence of DB underlies the therapeutic and side effects of this drug. However, the relevance of DB to the clinical actions of haloperidol has been questioned on the grounds that chronic drug-induced DB has not yet been demonstrated in freely moving animals. In this study, responding for rewarding electrical brain stimulation was used to assess the occurrence of DB in rats chronically treated with haloperidol or clozapine. The time course of the effects of acute haloperidol (7.8-500 microg/kg) and clozapine (5-40 mg/kg) and of withdrawal from chronic drug treatment on reward and performance measures were also characterized. Haloperidol and clozapine dose-dependently attenuated reward and performance, haloperidol producing a predominant suppression of performance, and clozapine preferentially attenuating reward. Chronic (21 d) treatment with haloperidol (500 microg/kg) caused responding to cease in the six rats tested, and repeated injection with apomorphine restored the behavior in all of them; such an effect of apomorphine was observed in only two of six rats treated acutely with the same dose of haloperidol. Chronic treatment with clozapine (20 mg/kg) increased reward thresholds, an effect that was reversed by apomorphine in chronically, but not acutely, treated rats. The times at which chronic haloperidol-treated rats resumed responding was positively correlated with indices of behavioral supersensitivity after withdrawal, suggesting that the effect of apomorphine was not caused by direct stimulation of upregulated postsynaptic receptors. These findings constitute the first behavioral evidence of DB in unanesthetized, freely moving animals treated chronically with antipsychotics. They also demonstrate that the neural substrates mediating reward and performance are functionally independent and differentially sensitive to haloperidol and clozapine.
Figures





References
-
- Akaoka H, Charléty P, Saunier C-F, Buda M, Chouvet G. Inhibition of nigral dopamine neurons by systemic and local apomorphine: possible contribution of dendritic autoreceptors. Neuroscience. 1992;49:879–891. - PubMed
-
- Barros HMT, Braz S, Carlini EA. Behavioural manifestations elicited by apomorphine, influence of the route of administration. Pharmacology. 1989;38:335–340. - PubMed
-
- Boye SM, Rompré P-P. Effect of pimozide on self-stimulation thresholds under a continuous and fixed-interval schedule of reinforcement. Behav Brain Res. 1996;78:243–245. - PubMed
-
- Bunney BS, Grace AA. Acute and chronic haloperidol treatment: comparison of effects on nigral dopaminergic cell activity. Life Sci. 1978;23:1715–1728. - PubMed
-
- Burt DR, Creese I, Snyder SH. Antischizophrenic drugs: chronic treatment elevates dopamine receptor binding in brain. Science. 1977;196:326–328. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
