A protein disulfide isomerase gene fusion expression system that increases the extracellular productivity of Bacillus brevis
- PMID: 10653729
- PMCID: PMC91874
- DOI: 10.1128/AEM.66.2.638-642.2000
A protein disulfide isomerase gene fusion expression system that increases the extracellular productivity of Bacillus brevis
Abstract
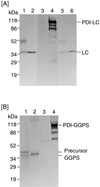
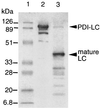
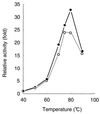
We have developed a versatile Bacillus brevis expression and secretion system based on the use of fungal protein disulfide isomerase (PDI) as a gene fusion partner. Fusion with PDI increased the extracellular production of heterologous proteins (light chain of immunoglobulin G, 8-fold; geranylgeranyl pyrophosphate synthase, 12-fold). Linkage to PDI prevented the aggregation of the secreted proteins, resulting in high-level accumulation of fusion proteins in soluble and biologically active forms. We also show that the disulfide isomerase activity of PDI in a fusion protein is responsible for the suppression of the aggregation of the protein with intradisulfide, whereas aggregation of the protein without intradisulfide was prevented even when the protein was fused to a mutant PDI whose two active sites were disrupted, suggesting that another PDI function, such as chaperone-like activity, synergistically prevented the aggregation of heterologous proteins in the PDI fusion expression system.
Figures


References
-
- Burnette W N. “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose, and radiographic detection with antibodies and radioiodinated protein A. Anal Biochem. 1981;112:195–203. - PubMed
-
- Gilbert H F. Protein disulfide isomerase and assisted protein folding. J Biol Chem. 1999;272:29399–29402. - PubMed
-
- Hayano T, Hirose M, Kikuchi M. Protein disulfide isomerase mutant lacking isomerase activity accelerates protein folding in the cell. FEBS Lett. 1995;377:505–511. - PubMed
-
- Hillson D A, Lambert N, Freedman R B. Formation and isomerization of disulfide bonds in proteins: protein disulfide-isomerase. Methods Enzymol. 1984;107:281–294. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

