Bovine herpesvirus 5 glycoprotein E is important for neuroinvasiveness and neurovirulence in the olfactory pathway of the rabbit
- PMID: 10666239
- PMCID: PMC111690
- DOI: 10.1128/jvi.74.5.2094-2106.2000
Bovine herpesvirus 5 glycoprotein E is important for neuroinvasiveness and neurovirulence in the olfactory pathway of the rabbit
Abstract
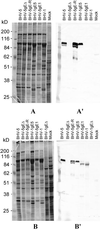
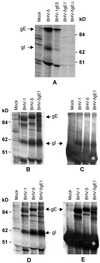
Glycoprotein E (gE) is important for full virulence potential of the alphaherpesviruses in both natural and laboratory hosts. The gE sequence of the neurovirulent bovine herpesvirus 5 (BHV-5) was determined and compared with that of the nonneurovirulent BHV-1. Alignment of the predicted amino acid sequences of BHV-1 and BHV-5 gE open reading frames showed that they had 72% identity and 77% similarity. To determine the role of gE in the differential neuropathogenesis of BHV-1 and BHV-5, we have constructed BHV-1 and BHV-5 recombinants: gE-deleted BHV-5 (BHV-5gEDelta), BHV-5 expressing BHV-1 gE (BHV-5gE1), and BHV-1 expressing BHV-5 gE (BHV-1gE5). Neurovirulence properties of these recombinant viruses were analyzed using a rabbit seizure model (S. I. Chowdhury et al., J. Comp. Pathol. 117:295-310, 1997) that distinguished wild-type BHV-1 and -5 based on their differential neuropathogenesis. Intranasal inoculation of BHV-5 gEDelta and BHV-5gE1 produced significantly reduced neurological signs that affected only 10% of the infected rabbits. The recombinant BHV-1gE5 did not invade the central nervous system (CNS). Virus isolation and immunohistochemistry data suggest that these recombinants replicate and spread significantly less efficiently in the brain than BHV-5 gE revertant or wild-type BHV-5, which produced severe neurological signs in 70 to 80% rabbits. Taken together, the results of neurological signs, brain lesions, virus isolation, and immunohistochemistry indicate that BHV-5 gE is important for efficient neural spread and neurovirulence within the CNS and could not be replaced by BHV-1 gE. However, BHV-5 gE is not required for initial viral entry into olfactory pathway.
Figures






Similar articles
-
Neurovirulence of glycoprotein C(gC)-deleted bovine herpesvirus type-5 (BHV-5) and BHV-5 expressing BHV-1 gC in a rabbit seizure model.J Neurovirol. 2000 Aug;6(4):284-95. doi: 10.3109/13550280009030754. J Neurovirol. 2000. PMID: 10951552
-
In the absence of glycoprotein I (gI), gE determines bovine herpesvirus type 5 neuroinvasiveness and neurovirulence.J Neurovirol. 2004 Aug;10(4):233-43. doi: 10.1080/13550280490463514. J Neurovirol. 2004. PMID: 15371153
-
A glycine-rich bovine herpesvirus 5 (BHV-5) gE-specific epitope within the ectodomain is important for BHV-5 neurovirulence.J Virol. 2004 May;78(9):4806-16. doi: 10.1128/jvi.78.9.4806-4816.2004. J Virol. 2004. PMID: 15078962 Free PMC article.
-
Infection and spread of alphaherpesviruses in the nervous system.Adv Virus Res. 1998;51:237-347. doi: 10.1016/s0065-3527(08)60787-3. Adv Virus Res. 1998. PMID: 9891589 Review. No abstract available.
-
Invasion of the Nervous System.Curr Issues Mol Biol. 2021;41:1-62. doi: 10.21775/cimb.041.001. Epub 2020 Jul 29. Curr Issues Mol Biol. 2021. PMID: 32723924 Review.
Cited by
-
Bovine herpesvirus 5 (BHV-5) Us9 is essential for BHV-5 neuropathogenesis.J Virol. 2002 Apr;76(8):3839-51. doi: 10.1128/jvi.76.8.3839-3851.2002. J Virol. 2002. PMID: 11907224 Free PMC article.
-
Comparative study on the in vitro and in vivo properties of two bovine herpesvirus-5 reference strains.Acta Vet Scand. 2011 Jun 8;53(1):37. doi: 10.1186/1751-0147-53-37. Acta Vet Scand. 2011. PMID: 21651813 Free PMC article.
-
Glycoprotein D of bovine herpesvirus 5 (BoHV-5) confers an extended host range to BoHV-1 but does not contribute to invasion of the brain.J Virol. 2010 Jun;84(11):5583-93. doi: 10.1128/JVI.00228-10. Epub 2010 Mar 10. J Virol. 2010. PMID: 20219909 Free PMC article.
-
Isolation and characterization of bovine herpes virus 5 (BoHV5) from cattle in India.PLoS One. 2020 Apr 24;15(4):e0232093. doi: 10.1371/journal.pone.0232093. eCollection 2020. PLoS One. 2020. PMID: 32330151 Free PMC article.
-
Envelope protein Us9 is required for the anterograde transport of bovine herpesvirus type 1 from trigeminal ganglia to nose and eye upon reactivation.J Neurovirol. 2007 Aug;13(4):384-8. doi: 10.1080/13550280701375433. J Neurovirol. 2007. PMID: 17849322
References
-
- Ashbaugh S E, Thompson K E, Belknap E B, Schultheiss P C, Chowdhury S I, Collins J K. Specific detection of shedding and latency of bovine herpesvirus 1 and 5 using a nested polymerase chain reaction. J Vet Diagn Investig. 1997;9:387–394. - PubMed
-
- Babic N, Klupp B, Brack A, Mettenleiter T C, Ugolini G, Flamand A. Deletion of glycoprotein gE reduces the propagation of pseudorabies virus in the nervous system of mice after intranasal inoculation. Virology. 1996;219:279–284. - PubMed
-
- Balan P, Davis-Poynter N, Bell S, Atkinson H, Browne H, Minson T. An analysis of the in vitro and in vivo phenotypes of mutants of herpes simplex virus type 1 lacking glycoproteins gG, gE, gI or the putative gJ. J Gen Virol. 1994;75:1245–1258. - PubMed
-
- Belknap E B, Collins J K, Ayers V K, Schulteiss P C. Experimental infection of neonatal calves with neurovirulent bovine herpes virus type 1.3. Vet Pathol. 1994;31:358–365. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

