Rotavirus infection induces an increase in intracellular calcium concentration in human intestinal epithelial cells: role in microvillar actin alteration
- PMID: 10666263
- PMCID: PMC111714
- DOI: 10.1128/jvi.74.5.2323-2332.2000
Rotavirus infection induces an increase in intracellular calcium concentration in human intestinal epithelial cells: role in microvillar actin alteration
Abstract
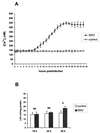
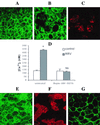
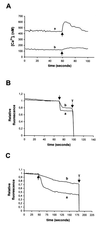
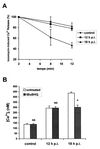
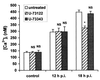
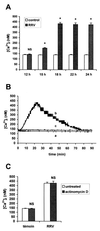
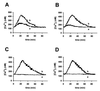
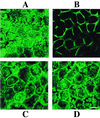
Rotaviruses, which infect mature enterocytes of the small intestine, are recognized as the most important cause of viral gastroenteritis in young children. We have previously reported that rotavirus infection induces microvillar F-actin disassembly in human intestinal epithelial Caco-2 cells (N. Jourdan, J. P. Brunet, C. Sapin, A. Blais, J. Cotte-Laffitte, F. Forestier, A. M. Quero, G. Trugnan, and A. L. Servin, J. Virol. 72:7228-7236, 1998). In this study, to determine the mechanism responsible for rotavirus-induced F-actin alteration, we investigated the effect of infection on intracellular calcium concentration ([Ca(2+)](i)) in Caco-2 cells, since Ca(2+) is known to be a determinant factor for actin cytoskeleton regulation. As measured by quin2 fluorescence, viral replication induced a progressive increase in [Ca(2+)](i) from 7 h postinfection, which was shown to be necessary and sufficient for microvillar F-actin disassembly. During the first hours of infection, the increase in [Ca(2+)](i) was related only to an increase in Ca(2+) permeability of plasmalemma. At a late stage of infection, [Ca(2+)](i) elevation was due to both extracellular Ca(2+) influx and Ca(2+) release from the intracellular organelles, mainly the endoplasmic reticulum (ER). We noted that at this time the [Ca(2+)](i) increase was partially related to a phospholipase C (PLC)-dependent mechanism, which probably explains the Ca(2+) release from the ER. We also demonstrated for the first time that viral proteins or peptides, released into culture supernatants of rotavirus-infected Caco-2 cells, induced a transient increase in [Ca(2+)](i) of uninfected Caco-2 cells, by a PLC-dependent efflux of Ca(2+) from the ER and by extracellular Ca(2+) influx. These supernatants induced a Ca(2+)-dependent microvillar F-actin alteration in uninfected Caco-2 cells, thus participating in rotavirus pathogenesis.
Figures
References
-
- Ball J M, Tian P, Zeng C Q-Y, Morris A P, Estes M K. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science. 1996;272:101–104. - PubMed
-
- Berridge M J. Inositol trisphosphate and calcium signalling. Nature. 1993;361:315–325. - PubMed
-
- Bishop R F, Davidson G P, Holmes I H, Ruck B J. Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet. 1973;ii:1281–1283. - PubMed
-
- Bleasdale J E, Thakur N R, Gremban R S, Bundy G L, Fitzpatrick F A, Smith R J, Bunting S. Selective inhibition of receptor-coupled phospholipase C-dependent processes in human platelets and polymorphonuclear neutrophils. J Pharmacol Exp Ther. 1990;255:756–768. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

