A stable HeLa cell line that inducibly expresses poliovirus 2A(pro): effects on cellular and viral gene expression
- PMID: 10666269
- PMCID: PMC111720
- DOI: 10.1128/jvi.74.5.2383-2392.2000
A stable HeLa cell line that inducibly expresses poliovirus 2A(pro): effects on cellular and viral gene expression
Abstract
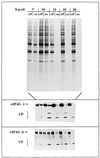
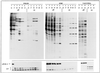
A HeLa cell clone (2A7d) that inducibly expresses the gene for poliovirus protease 2A (2A(pro)) under the control of tetracycline has been obtained. Synthesis of 2A(pro) induces severe morphological changes in 2A7d cells. One day after tetracycline removal, cells round up and a few hours later die. Poliovirus 2A(pro) cleaves both forms of initiation factor eIF4G, causing extensive inhibition of capped-mRNA translation a few hours after protease induction. Methoxysuccinyl-Ala-Ala-Pro-Val-chloromethylketone, a selective inhibitor of 2A(pro), prevents both eIF4G cleavage and inhibition of translation but not cellular death. Expression of 2A(pro) still allows both the replication of poliovirus and the translation of mRNAs containing a picornavirus leader sequence, while vaccinia virus replication is drastically inhibited. Translation of transfected capped mRNA is blocked in 2A7d-On cells, while luciferase synthesis from a mRNA bearing a picornavirus internal ribosome entry site (IRES) sequence is enhanced by the presence of 2A(pro). Moreover, synthesis of 2A(pro) in 2A7d cells complements the translational defect of a poliovirus 2A(pro)-defective variant. These results show that poliovirus 2A(pro) expression mimics some phenotypical characteristics of poliovirus-infected cells, such as cell rounding, inhibition of protein synthesis and enhancement of IRES-driven translation. This cell line constitutes a useful tool to further analyze 2A(pro) functions, to complement poliovirus 2A(pro) mutants, and to test antiviral compounds.
Figures




References
-
- Aldabe R, Carrasco L. Induction of membrane proliferation by poliovirus proteins 2C and 2BC. Biochem Biophys Res Commun. 1995;206:64–76. - PubMed
-
- Aldabe R, Feduchi E, Novoa I, Carrasco L. Expression of poliovirus 2Apro in mammalian cells: effects on translation. FEBS Lett. 1995;377:1–5. - PubMed
-
- Aldabe R, Feduchi E, Novoa I, Carrasco L. Efficient cleavage of p220 by poliovirus 2Apro expression in mammalian cells: effects on vaccinia virus. Biochem Biophys Res Commun. 1995;215:928–936. - PubMed
-
- Aranda M, Maule A. Virus-induced host gene shutoff in animals and plants. Virology. 1998;243:261–267. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

