Complex protein interactions within the human polyadenylation machinery identify a novel component
- PMID: 10669729
- PMCID: PMC85326
- DOI: 10.1128/MCB.20.5.1515-1525.2000
Complex protein interactions within the human polyadenylation machinery identify a novel component
Abstract
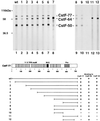
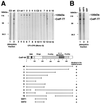
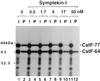
Polyadenylation of mRNA precursors is a two-step reaction requiring multiple protein factors. Cleavage stimulation factor (CstF) is a heterotrimer necessary for the first step, endonucleolytic cleavage, and it plays an important role in determining the efficiency of polyadenylation. Although a considerable amount is known about the RNA binding properties of CstF, the protein-protein interactions required for its assembly and function are poorly understood. We therefore first identified regions of the CstF subunits, CstF-77, CstF-64, and CstF-50, required for interaction with each other. Unexpectedly, small regions of two of the subunits participate in multiple interactions. In CstF-77, a proline-rich domain is necessary not only for binding both other subunits but also for self-association, an interaction consistent with genetic studies in Drosophila. In CstF-64, a small region, highly conserved in metazoa, is responsible for interactions with two proteins, CstF-77 and symplekin, a nuclear protein of previously unknown function. Intriguingly, symplekin has significant similarity to a yeast protein, PTA1, that is a component of the yeast polyadenylation machinery. We show that multiple factors, including CstF, cleavage-polyadenylation specificity factor, and symplekin, can be isolated from cells as part of a large complex. These and other data suggest that symplekin may function in assembly of the polyadenylation machinery.
Figures



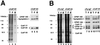
References
-
- Beyer K, Dandekar T, Keller W. RNA ligands selected by cleavage stimulation factor contain distinct sequence motifs that function as downstream elements in 3′-end processing of pre-mRNA. J Biol Chem. 1997;272:26769–26779. - PubMed
-
- Colgan D F, Manley J L. Mechanism and regulation of mRNA polyadenylation. Genes Dev. 1997;11:2755–2766. - PubMed
-
- Dantonel J-C, Murthy K G K, Manley J L, Tora L. CPSF links transcription and mRNA 3′ end formation. Nature. 1997;389:399–402. - PubMed
-
- Faber P W, Barnes G T, Srinidhi J, Chen J, Gusella J F, MacDonald M E. Huntingtin interacts with a family of WW domain proteins. Hum Mol Genet. 1998;7:1463–1474. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
