Control of human telomere length by TRF1 and TRF2
- PMID: 10669743
- PMCID: PMC85349
- DOI: 10.1128/MCB.20.5.1659-1668.2000
Control of human telomere length by TRF1 and TRF2
Abstract
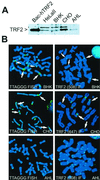
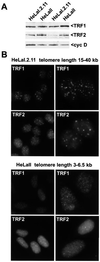
Telomere length in human cells is controlled by a homeostasis mechanism that involves telomerase and the negative regulator of telomere length, TRF1 (TTAGGG repeat binding factor 1). Here we report that TRF2, a TRF1-related protein previously implicated in protection of chromosome ends, is a second negative regulator of telomere length. Overexpression of TRF2 results in the progressive shortening of telomere length, similar to the phenotype observed with TRF1. However, while induction of TRF1 could be maintained over more than 300 population doublings and resulted in stable, short telomeres, the expression of exogenous TRF2 was extinguished and the telomeres eventually regained their original length. Consistent with their role in measuring telomere length, indirect immunofluorescence indicated that both TRF1 and TRF2 bind to duplex telomeric DNA in vivo and are more abundant on telomeres with long TTAGGG repeat tracts. Neither TRF1 nor TRF2 affected the expression level of telomerase. Furthermore, the presence of TRF1 or TRF2 on a short linear telomerase substrate did not inhibit the enzymatic activity of telomerase in vitro. These findings are consistent with the recently proposed t loop model of telomere length homeostasis in which telomerase-dependent telomere elongation is blocked by sequestration of the 3' telomere terminus in TRF1- and TRF2-induced telomeric loops.
Figures



References
-
- Ashley T, Ward D C. A “hot spot” of recombination coincides with an interstitial telomeric sequence in the Armenian hamster. Cytogenet Cell Genet. 1993;62:169–171. - PubMed
-
- Auffray C, Rougeon G. Purification of mouse immunoglobulin heavy-chain messenger RNAs from total myeloma tumor RNA. Eur J Biochem. 1980;107:303–314. - PubMed
-
- Bianchi A. Characterization of DNA binding activities at vertebrate telomeres. Doctoral dissertation. New York, N.Y: The Rockefeller University; 1999.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
