Arrest of G(1)-S progression by the p53-inducible gene PC3 is Rb dependent and relies on the inhibition of cyclin D1 transcription
- PMID: 10669755
- PMCID: PMC85361
- DOI: 10.1128/MCB.20.5.1797-1815.2000
Arrest of G(1)-S progression by the p53-inducible gene PC3 is Rb dependent and relies on the inhibition of cyclin D1 transcription
Abstract
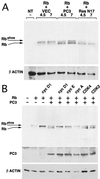
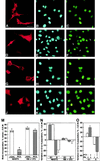
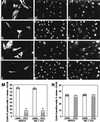
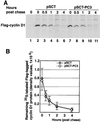
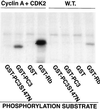
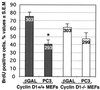
The p53-inducible gene PC3 (TIS21, BTG2) is endowed with antiproliferative activity. Here we report that expression of PC3 in cycling cells induced accumulation of hypophosphorylated, growth-inhibitory forms of pRb and led to G(1) arrest. This latter was not observed in cells with genetic disruption of the Rb gene, indicating that the PC3-mediated G(1) arrest was Rb dependent. Furthermore, (i) the arrest of G(1)-S transition exerted by PC3 was completely rescued by coexpression of cyclin D1 but not by that of cyclin A or E; (ii) expression of PC3 caused a significant down-regulation of cyclin D1 protein levels, also in Rb-defective cells, accompanied by inhibition of CDK4 activity in vivo; and (iii) the removal from the PC3 molecule of residues 50 to 68, a conserved domain of the PC3/BTG/Tob gene family, which we term GR, led to a loss of the inhibition of proliferation as well as of the down-regulation of cyclin D1 levels. These data point to cyclin D1 down-regulation as the main factor responsible for the growth inhibition by PC3. Such an effect was associated with a decrease of cyclin D1 transcript and of cyclin D1 promoter activity, whereas no effect of PC3 was observed on cyclin D1 protein stability. Taken together, these findings indicate that PC3 impairs G(1)-S transition by inhibiting pRb function in consequence of a reduction of cyclin D1 levels and that PC3 acts, either directly or indirectly, as a transcriptional regulator of cyclin D1.
Figures








Similar articles
-
The gene PC3(TIS21/BTG2), prototype member of the PC3/BTG/TOB family: regulator in control of cell growth, differentiation, and DNA repair?J Cell Physiol. 2001 May;187(2):155-65. doi: 10.1002/jcp.1062. J Cell Physiol. 2001. PMID: 11267995 Review.
-
TIS21/BTG2/PC3 and cyclin D1 are key determinants of nuclear diacylglycerol kinase-zeta-dependent cell cycle arrest.Cell Signal. 2009 May;21(5):801-9. doi: 10.1016/j.cellsig.2009.01.027. Epub 2009 Jan 20. Cell Signal. 2009. PMID: 19263516
-
HDAC1, HDAC4, and HDAC9 Bind to PC3/Tis21/Btg2 and Are Required for Its Inhibition of Cell Cycle Progression and Cyclin D1 Expression.J Cell Physiol. 2017 Jul;232(7):1696-1707. doi: 10.1002/jcp.25467. Epub 2017 Jan 27. J Cell Physiol. 2017. PMID: 27333946
-
Acute renal failure during sepsis: potential role of cell cycle regulation.J Infect. 2009 Jun;58(6):459-64. doi: 10.1016/j.jinf.2009.04.003. Epub 2009 Apr 17. J Infect. 2009. PMID: 19428114
-
Genetic instability as a consequence of inappropriate entry into and progression through S-phase.Cancer Metastasis Rev. 1995 Mar;14(1):59-73. doi: 10.1007/BF00690212. Cancer Metastasis Rev. 1995. PMID: 7606822 Review.
Cited by
-
An anxiogenic drug, FG 7142, induced an increase in mRNA of Btg2 and Adamts1 in the hippocampus of adult mice.Behav Brain Funct. 2012 Aug 22;8:43. doi: 10.1186/1744-9081-8-43. Behav Brain Funct. 2012. PMID: 22913326 Free PMC article.
-
Tumor suppressors BTG1 and BTG2: Beyond growth control.J Cell Physiol. 2019 May;234(5):5379-5389. doi: 10.1002/jcp.27407. Epub 2018 Oct 23. J Cell Physiol. 2019. PMID: 30350856 Free PMC article. Review.
-
Response of rat muscle to acute resistance exercise defined by transcriptional and translational profiling.J Physiol. 2002 Nov 15;545(1):27-41. doi: 10.1113/jphysiol.2002.021220. J Physiol. 2002. PMID: 12433947 Free PMC article.
-
Transducer of ERBB2.1 (TOB1) as a Tumor Suppressor: A Mechanistic Perspective.Int J Mol Sci. 2015 Dec 15;16(12):29815-28. doi: 10.3390/ijms161226203. Int J Mol Sci. 2015. PMID: 26694352 Free PMC article. Review.
-
Cucurbitacin B Inhibits the Proliferation of WPMY-1 Cells and HPRF Cells via the p53/MDM2 Axis.Int J Mol Sci. 2024 Aug 28;25(17):9333. doi: 10.3390/ijms25179333. Int J Mol Sci. 2024. PMID: 39273281 Free PMC article.
References
-
- Agarwal M L, Taylor W R, Chernow M V, Chernova O B, Stark G R. The p53 network. J Biol Chem. 1998;273:1–4. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
