Akt promotes survival of cardiomyocytes in vitro and protects against ischemia-reperfusion injury in mouse heart
- PMID: 10673259
- PMCID: PMC3627349
- DOI: 10.1161/01.cir.101.6.660
Akt promotes survival of cardiomyocytes in vitro and protects against ischemia-reperfusion injury in mouse heart
Abstract
Background: IGF-1 has been shown to protect myocardium against death in animal models of infarct and ischemia-reperfusion injury. In the present study, we investigated the role of the IGF-1-regulated protein kinase Akt in cardiac myocyte survival in vitro and in vivo.
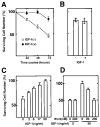
Methods and results: IGF-1 promoted survival of cultured cardiomyocytes under conditions of serum deprivation in a dose-dependent manner but had no effect on cardiac fibroblast survival. The cytoprotective effect of IGF-1 on cardiomyocytes was abrogated by the phosphatidylinositol 3-kinase (PI 3-kinase) inhibitor wortmannin. Wortmannin had no effect on cardiomyocyte viability in the absence of IGF-1. IGF-1-mediated cytoprotection correlated with the wortmannin-sensitive induction of Akt protein kinase activity. To examine the functional consequences of Akt activation in cardiomyocyte survival, replication-defective adenoviral constructs expressing wild-type, dominant-negative, and constitutively active Akt genes were constructed. Transduction of dominant-negative Akt blocked IGF-1-induced survival but had no effect on cardiomyocyte survival in the absence of IGF-1. In contrast, transduction of wild-type Akt enhanced cardiomyocyte survival at subsaturating levels of IGF-1, whereas constitutively active Akt protected cardiomyocytes from apoptosis in the absence of IGF-1. After transduction into the mouse heart in vivo, constitutively active Akt protected against myocyte apoptosis in response to ischemia-reperfusion injury.
Conclusions: These data are the first documentation that Akt functions to promote cellular survival in vivo, and they indicate that the activation of this pathway may be useful in promoting myocyte survival in the diseased heart.
Figures


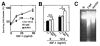


References
-
- Kajstura J, Cheng W, Reiss K, Clark WA, Sonnenblick EH, Krajewski S, Reed JC, Olivetti G, Anversa P. Apoptotic and necrotic cell deaths are independent contributing variables of infarct size in rats. Lab Invest. 1996;74:86–107. - PubMed
-
- Soonpaa MH, Field LJ. Survey of studies examining mammalian cardiomyocyte DNA synthesis. Circ Res. 1998;83:15–26. - PubMed
-
- Anversa P, Kajstura J. Ventricular myocytes are not terminally differentiated in the adult mammalian heart. Circ Res. 1998;83:1–14. - PubMed
-
- MacLellan WR, Schneider MD. Death by design: programmed cell death in cardiovascular biology and disease. Circ Res. 1997;81:137–144. - PubMed
-
- Hirota H, Chen J, Betz U, Rajewsky K, Gu Y, Ross J, Jr, Muller W, Chien K. Loss of a gp130 cardiac muscle cell survival pathway is a critical event in the onset of heart failure during biomechanical stress. Cell. 1999;97:189–198. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

