Iron deficiency decreases the Fe(III)-chelate reducing activity of leaf protoplasts
- PMID: 10677427
- PMCID: PMC58871
- DOI: 10.1104/pp.122.2.337
Iron deficiency decreases the Fe(III)-chelate reducing activity of leaf protoplasts
Abstract
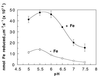
The ferric-chelate reductase (FC-R) activity of mesophyll protoplasts isolated from Fe-sufficient (control) and Fe-deficient sugar beet (Beta vulgaris L.) leaves has been characterized. Measurements were made in an ionic environment similar to that in the apoplastic space of the sugar beet mesophyll cells. The FC-R activity of Fe-sufficient and Fe-deficient protoplasts was dependent on light. Fe deficiency decreased markedly the FC-R activity per protoplast surface unit. The optimal pH for the activity of the FC-R in mesophyll protoplasts was in the range 5.5 to 6.0, typical of the apoplastic space. Beyond pH 6.0, the activity of the FC-R in mesophyll protoplasts decreased markedly in both Fe-sufficient and Fe-deficient protoplasts. These data suggest that both the intrinsic decrease in FC-R activity per protoplast surface and a possible shift in the pH of the apoplastic space could lead to the accumulation of physiologically inactive Fe pools in chlorotic leaves.
Figures

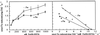
Similar articles
-
Technical advance: reduction of Fe(III)-chelates by mesophyll leaf disks of sugar beet. Multi-component origin and effects of Fe deficiency.Plant Cell Physiol. 2001 Jan;42(1):94-105. doi: 10.1093/pcp/pce012. Plant Cell Physiol. 2001. PMID: 11158448
-
Changes in iron and organic acid concentrations in xylem sap and apoplastic fluid of iron-deficient Beta vulgaris plants in response to iron resupply.J Plant Physiol. 2010 Mar 1;167(4):255-60. doi: 10.1016/j.jplph.2009.09.007. Epub 2009 Oct 24. J Plant Physiol. 2010. PMID: 19854536
-
Characterization of FRO1, a pea ferric-chelate reductase involved in root iron acquisition.Plant Physiol. 2002 May;129(1):85-94. doi: 10.1104/pp.010829. Plant Physiol. 2002. PMID: 12011340 Free PMC article.
-
Towards a knowledge-based correction of iron chlorosis.Plant Physiol Biochem. 2011 May;49(5):471-82. doi: 10.1016/j.plaphy.2011.01.026. Epub 2011 Feb 3. Plant Physiol Biochem. 2011. PMID: 21349731 Review.
-
Ferric iron reduction and iron uptake in eucaryotes: studies with the yeasts Saccharomyces cerevisiae and Schizosaccharomyces pombe.Adv Exp Med Biol. 1994;356:81-9. doi: 10.1007/978-1-4615-2554-7_9. Adv Exp Med Biol. 1994. PMID: 7887248 Review. No abstract available.
Cited by
-
Physiological and biochemical adjustment of iron chlorosis affected low-chill peach cultivars supplied with different iron sources.Physiol Mol Biol Plants. 2012 Apr;18(2):141-8. doi: 10.1007/s12298-012-0107-9. Epub 2012 Apr 3. Physiol Mol Biol Plants. 2012. PMID: 23573051 Free PMC article.
-
Protein profile of Beta vulgaris leaf apoplastic fluid and changes induced by Fe deficiency and Fe resupply.Front Plant Sci. 2015 Mar 18;6:145. doi: 10.3389/fpls.2015.00145. eCollection 2015. Front Plant Sci. 2015. PMID: 25852707 Free PMC article.
-
Regulation of Iron Homeostasis and Use in Chloroplasts.Int J Mol Sci. 2020 May 11;21(9):3395. doi: 10.3390/ijms21093395. Int J Mol Sci. 2020. PMID: 32403383 Free PMC article. Review.
-
Rootstock influence on iron uptake responses in Citrus leaves and their regulation under the Fe paradox effect.PeerJ. 2017 Sep 25;5:e3553. doi: 10.7717/peerj.3553. eCollection 2017. PeerJ. 2017. PMID: 28966887 Free PMC article.
-
Light-regulated, tissue-specific, and cell differentiation-specific expression of the Arabidopsis Fe(III)-chelate reductase gene AtFRO6.Plant Physiol. 2006 Apr;140(4):1345-54. doi: 10.1104/pp.105.074138. Epub 2006 Feb 17. Plant Physiol. 2006. PMID: 16489134 Free PMC article.
References
-
- Abadía J. Leaf responses to Fe deficiency: a review. J Plant Nutr. 1992;15:1699–1713.
-
- Abadía J, Abadía A. Iron and plant pigments. In: Barton LL, Hemming BC, editors. Iron Chelation in Plants and Soil Microorganisms. New York: Academic Press; 1993. pp. 327–343.
-
- Abadía J, Monge E, Montañés L, Heras L. Extraction of iron from plant leaves by Fe (II) chelators. J Plant Nutr. 1984;7:777–784.
-
- Bienfait HF. Regulated redox processes at the plasmalemma of plant root cells and their function in iron uptake. J Bioenerg Biomembr. 1985;17:73–83. - PubMed
-
- Bienfait HF. Mechanisms in Fe-efficiency reactions of higher plants. J Plant Nutr. 1988;11:605–629.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

