Chemical chaperones mediate increased secretion of mutant alpha 1-antitrypsin (alpha 1-AT) Z: A potential pharmacological strategy for prevention of liver injury and emphysema in alpha 1-AT deficiency
- PMID: 10677536
- PMCID: PMC26515
- DOI: 10.1073/pnas.97.4.1796
Chemical chaperones mediate increased secretion of mutant alpha 1-antitrypsin (alpha 1-AT) Z: A potential pharmacological strategy for prevention of liver injury and emphysema in alpha 1-AT deficiency
Abstract
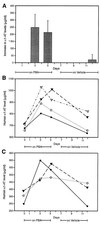
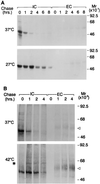
In alpha1-AT deficiency, a misfolded but functionally active mutant alpha1-ATZ (alpha1-ATZ) molecule is retained in the endoplasmic reticulum of liver cells rather than secreted into the blood and body fluids. Emphysema is thought to be caused by the lack of circulating alpha1-AT to inhibit neutrophil elastase in the lung. Liver injury is thought to be caused by the hepatotoxic effects of the retained alpha1-ATZ. In this study, we show that several "chemical chaperones," which have been shown to reverse the cellular mislocalization or misfolding of other mutant plasma membrane, nuclear, and cytoplasmic proteins, mediate increased secretion of alpha1-ATZ. In particular, 4-phenylbutyric acid (PBA) mediated a marked increase in secretion of functionally active alpha1-ATZ in a model cell culture system. Moreover, oral administration of PBA was well tolerated by PiZ mice (transgenic for the human alpha1-ATZ gene) and consistently mediated an increase in blood levels of human alpha1-AT reaching 20-50% of the levels present in PiM mice and normal humans. Because clinical studies have suggested that only partial correction is needed for prevention of both liver and lung injury in alpha1-AT deficiency and PBA has been used safely in humans, it constitutes an excellent candidate for chemoprophylaxis of target organ injury in alpha1-AT deficiency.
Figures


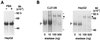
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

