M-type K+ currents in rat cultured thoracolumbar sympathetic neurones and their role in uracil nucleotide-evoked noradrenaline release
- PMID: 10683196
- PMCID: PMC1571887
- DOI: 10.1038/sj.bjp.0703096
M-type K+ currents in rat cultured thoracolumbar sympathetic neurones and their role in uracil nucleotide-evoked noradrenaline release
Abstract
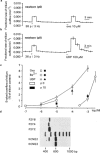
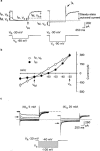
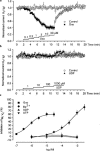
Cultured sympathetic neurones are depolarized and release noradrenaline in response to extracellular ATP, UDP and UTP. We examined the possibility that, in neurones cultured from rat thoracolumbar sympathetic ganglia, inhibition of the M-type potassium current might underlie the effects of UDP and UTP. Reverse transcriptase-polymerase chain reaction indicated that the cultured cells contained mRNA for P2Y(2)-, P2Y(4)- and P2Y(6)-receptors as well as for the KCNQ2- and KCNQ3-subunits which have been suggested to assemble into M-channels. In cultures of neurones taken from newborn as well as from 10 day-old rats, oxotremorine, the M-channel blocker Ba(2+) and UDP all released previously stored [(3)H]-noradrenaline. The neurones possessed M-currents, the kinetic properties of which were similar in neurones from newborn and 9 - 12 day-old rats. UDP, UTP and ATP had no effect on M-currents in neurones prepared from newborn rats. Oxotremorine and Ba(2+) substantially inhibited the current. ATP also had no effect on the M-current in neurones prepared from 9 - 12 day-old rats. Oxotremorine and Ba(2+) again caused marked inhibition. In contrast to cultures from newborn animals, UDP and UTP attenuated the M-current in neurones from 9 - 12 day-old rats; however, the maximal inhibition was less than 30%. The results indicate that inhibition of the M-current is not involved in uracil nucleotide-induced transmitter release from rat cultured sympathetic neurones during early development. M-current inhibition may contribute to release at later stages, but only to a minor extent. The mechanism leading to noradrenaline release by UDP and UTP remains unknown.
Figures




Similar articles
-
Role of action potentials and calcium influx in ATP- and UDP-induced noradrenaline release from rat cultured sympathetic neurones.Naunyn Schmiedebergs Arch Pharmacol. 1999 May;359(5):360-9. doi: 10.1007/pl00005362. Naunyn Schmiedebergs Arch Pharmacol. 1999. PMID: 10498285
-
Stimulation of mouse cultured sympathetic neurons by uracil but not adenine nucleotides.Neuroscience. 2001;103(1):227-36. doi: 10.1016/s0306-4522(00)00547-9. Neuroscience. 2001. PMID: 11311803
-
Autoinhibition of transmitter release from PC12 cells and sympathetic neurons through a P2Y receptor-mediated inhibition of voltage-gated Ca2+ channels.Eur J Neurosci. 2004 Dec;20(11):2917-28. doi: 10.1111/j.1460-9568.2004.03760.x. Eur J Neurosci. 2004. PMID: 15579146
-
Inhibition of potassium and calcium currents in neurones by molecularly-defined P2Y receptors.J Auton Nerv Syst. 2000 Jul 3;81(1-3):31-6. doi: 10.1016/s0165-1838(00)00150-8. J Auton Nerv Syst. 2000. PMID: 10869697 Review.
-
P2-purinoceptors on postganglionic sympathetic neurones.J Auton Pharmacol. 1996 Dec;16(6):413-6. doi: 10.1111/j.1474-8673.1996.tb00065.x. J Auton Pharmacol. 1996. PMID: 9131428 Review.
Cited by
-
Interaction of P2 purinergic receptors with cellular macromolecules.Naunyn Schmiedebergs Arch Pharmacol. 2008 Mar;377(1):1-33. doi: 10.1007/s00210-007-0222-2. Epub 2007 Dec 19. Naunyn Schmiedebergs Arch Pharmacol. 2008. PMID: 18273661 Free PMC article. Review.
-
P2Y receptor regulation of cultured rat cerebral cortical cells: calcium responses and mRNA expression in neurons and glia.Br J Pharmacol. 2003 May;139(2):279-88. doi: 10.1038/sj.bjp.0705242. Br J Pharmacol. 2003. PMID: 12770933 Free PMC article.
-
Mechanisms of sympathoexcitation via P2Y6 receptors.Front Pharmacol. 2022 Nov 3;13:1014284. doi: 10.3389/fphar.2022.1014284. eCollection 2022. Front Pharmacol. 2022. PMID: 36408258 Free PMC article.
-
Regulation of neuronal ion channels via P2Y receptors.Purinergic Signal. 2004 Dec;1(1):31-41. doi: 10.1007/s11302-004-4746-3. Purinergic Signal. 2004. PMID: 18404398 Free PMC article.
-
Functions of neuronal P2Y receptors.Pflugers Arch. 2006 Aug;452(5):538-51. doi: 10.1007/s00424-006-0063-8. Epub 2006 May 10. Pflugers Arch. 2006. PMID: 16691392 Review.
References
-
- ADAMS D.J., HARPER A.A.Electrophysiological properties of autonomic ganglion neurons Autonomic ganglia 19956Harwood Academic Publishers: Luxembourg; 153–212.In: Mc Lachlan, E.M.(ed.) Autonomic Nervous System Series
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

