Adverse effects of an active fragment of parathyroid hormone on rat hippocampal organotypic cultures
- PMID: 10694198
- PMCID: PMC1621111
- DOI: 10.1038/sj.bjp.0702949
Adverse effects of an active fragment of parathyroid hormone on rat hippocampal organotypic cultures
Abstract
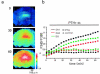
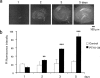
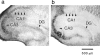
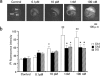
Adverse effects of an active fragment of parathyroid hormone (PTH(1 - 34)), a blood Ca(2+) level-regulating hormone, were examined using rat hippocampal slices in organotypic culture. Exposure of cultured slice preparations to 0.1 microM PTH(1 - 34) for 60 min resulted in a gradual increase in the intracellular Ca(2+) concentration ([Ca(2+)](i)); this effect was most obvious in the apical dendritic region of CA1 subfield. When PTH(1 - 34) at a lower concentration (1 nM) was added to the culture medium and its toxic effects examined using a propidium iodide intercalation method, significant toxicity was seen 3 days after exposure and increased with time. Cells in the CA1 region seemed more vulnerable to the hormone than cells in other regions. At 1 week of exposure, the toxic effects were dose-dependent over the range of 0.1 pM to 0.1 microM, the minimum effective dose being 10 pM. The adverse effects were not induced either by the inactive fragment, PTH(39 - 84), or by an active fragment of PTH-related peptide (PTHrP(1 - 34)), an intrinsic ligand of the brain PTH receptor. The PTH(1 - 34)-induced adverse effects were significantly inhibited by co-administration of 10 microM nifedipine, an L-type Ca(2+) channel blocker, but not by co-administration of blockers of the other types of Ca(2+) channel. The present study demonstrates that sustained high levels of PTH in the brain might cause degeneration of specific brain regions due to Ca(2+) overloading via activation of dihydropyridine-sensitive Ca(2+) channels, and suggests that PTH may be a risk factor for senile dementia. British Journal of Pharmacology (2000) 129, 21 - 28
Figures

Similar articles
-
Activation of dihydropyridine sensitive Ca2+ channels in rat hippocampal neurons in culture by parathyroid hormone.Neurosci Lett. 1998 Nov 13;256(3):139-42. doi: 10.1016/s0304-3940(98)00782-4. Neurosci Lett. 1998. PMID: 9855359
-
Expression of parathyroid hormone-related peptide (PTH-rp) and its receptorin the porcine ovary: regulation by transforming growth factor-beta and possible paracrine effects of granulosa cell PTH-rp secretion on theca cells.Biol Reprod. 2000 Feb;62(2):334-9. doi: 10.1095/biolreprod62.2.334. Biol Reprod. 2000. PMID: 10642570
-
Overexpression of regucalcin enhances its nuclear localization and suppresses L-type Ca2+ channel and calcium-sensing receptor mRNA expressions in cloned normal rat kidney proximal tubular epithelial NRK52E cells.J Cell Biochem. 2006 Nov 1;99(4):1064-77. doi: 10.1002/jcb.20863. J Cell Biochem. 2006. PMID: 16767692
-
1alpha(OH)D3 One-alpha-hydroxy-cholecalciferol--an active vitamin D analog. Clinical studies on prophylaxis and treatment of secondary hyperparathyroidism in uremic patients on chronic dialysis.Dan Med Bull. 2008 Nov;55(4):186-210. Dan Med Bull. 2008. PMID: 19232159 Review.
-
Defects in B-cell function and metabolism in uremia: role of parathyroid hormone.Kidney Int Suppl. 2001 Feb;78:S186-9. doi: 10.1046/j.1523-1755.2001.59780186.x. Kidney Int Suppl. 2001. PMID: 11169008 Review.
Cited by
-
Class II G protein-coupled receptors and their ligands in neuronal function and protection.Neuromolecular Med. 2005;7(1-2):3-36. doi: 10.1385/nmm:7:1-2:003. Neuromolecular Med. 2005. PMID: 16052036 Free PMC article. Review.
-
The vitamin D, ionised calcium and parathyroid hormone axis of cerebral capillary function: therapeutic considerations for vascular-based neurodegenerative disorders.PLoS One. 2015 Apr 13;10(4):e0125504. doi: 10.1371/journal.pone.0125504. eCollection 2015. PLoS One. 2015. PMID: 25874538 Free PMC article.
-
Fall prevention and vitamin D in the elderly: an overview of the key role of the non-bone effects.J Neuroeng Rehabil. 2010 Oct 11;7:50. doi: 10.1186/1743-0003-7-50. J Neuroeng Rehabil. 2010. PMID: 20937091 Free PMC article. Review.
-
Chemical modification of class II G protein-coupled receptor ligands: frontiers in the development of peptide analogs as neuroendocrine pharmacological therapies.Pharmacol Ther. 2010 Jan;125(1):39-54. doi: 10.1016/j.pharmthera.2009.07.006. Epub 2009 Aug 15. Pharmacol Ther. 2010. PMID: 19686775 Free PMC article. Review.
-
Aspartame and Soft Drink-Mediated Neurotoxicity in Rats: Implication of Oxidative Stress, Apoptotic Signaling Pathways, Electrolytes and Hormonal Levels.Metab Brain Dis. 2017 Oct;32(5):1639-1647. doi: 10.1007/s11011-017-0052-y. Epub 2017 Jun 28. Metab Brain Dis. 2017. PMID: 28660358
References
-
- ABOU-SAMURA A.B., JUPPNER H., FORCE T., FREEMAN M.W., KONG X.F., SCHIPANI E., URENA P., RICHARDS J., BONVENTRE J.V., POTTS J.T. Expression cloning of a common receptor for parathyroid hormone and parathyroid hormone-related peptide from rat osteoblast-like cells; a single receptor stimulates intracellular accumulation of both cAMP and inositol triphosphates and increases intracellular free calcium. Proc. Natl. Acad. Sci. U.S.A. 1992;89:2732–2736. - PMC - PubMed
-
- BACSKAI B.J., FRIEDMAN P.A. Activation of latent Ca2+ channels in renal epithelial cells by parathyroid hormone. Nature. 1990;347:388–391. - PubMed
-
- BEHAR V., NAKAMOTO C., GREENBER Z. , BISELLO B, A. , SUVA L.J., ROSENBLATT M., CHOREV M. Histidine at position 5 is the specificity ‘switch' between two parathyroid hormone receptor subtypes. Endocrinology. 1996;137:4217–4224. - PubMed
-
- CHOI D.W. Calcium-mediated neurotoxicity: relationship to specific channel types and role in ischemic damage. Trends Neurosci. 1988;7:369–371. - PubMed
-
- DISTERHORFT J.F., MOYER J.R., Jr, THOMPSON L.T. The calcium rationale in aging and Alzheimer's disease. Evidence from an animal model of normal aging. Ann. N.Y. Acad. Sci. 1994;747:382–406. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

