Stimulation of the ventral tegmental area enhances the effect of vasopressin on blood pressure in conscious rats
- PMID: 10694199
- PMCID: PMC1621113
- DOI: 10.1038/sj.bjp.0702982
Stimulation of the ventral tegmental area enhances the effect of vasopressin on blood pressure in conscious rats
Abstract
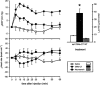
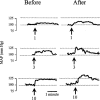
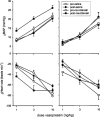
The mesolimbic dopamine system projects to a large number of forebrain areas and plays an important role in the regulation of locomotor activity, cognition and reward. We previously found evidence for a functional interaction between the mesolimbic dopamine system and circulating vasopressin and the present study was performed to test the hypothesis that mesolimbic dopamine stimulation modulates the cardiovascular effects of vasopressin. Sprague-Dawley rats were stereotaxically implanted with a guide cannula into the region of origin of the mesolimbic system, the ventral tegmental area, and instrumented with catheters into the abdominal aorta and jugular vein. One week later, separate groups of conscious rats were injected intravenously with 1, 3 or 10 ng kg(-1) of arginine-vasopressin or other vasopressor drugs before and after intra-ventral tegmental area injection of 10 nmol of neurotensin. Intra-ventral tegmental area injections of neurotensin had no significant effect on mean arterial pressure and heart rate but significantly potentiated the pressor response to intravenous administration of vasopressin when compared to saline-injections. However, the vasopressin-induced bradycardia was unaffected. Intravenous pretreatment with raclopride blocked the ability of neurotensin, injected into the ventral tegmental area, to potentiate the vasopressin-induced pressor response. Intra ventral tegmental area injections of neurotensin had no effect on the pressor response and bradycardia induced by intravenous angiotensin II or methoxamine. In conclusion, these results suggest that the mesolimbic dopamine system, in addition to its well-known role in the regulation of behaviour, modulates cardiovascular control by potentiating the effects of vasopressin on mean arterial pressure. British Journal of Pharmacology (2000) 129, 29 - 36
Figures
Similar articles
-
A functional interaction between the mesolimbic dopamine system and vasopressin release in the regulation of blood pressure in conscious rats.Neuroscience. 1997 Nov;81(1):69-78. doi: 10.1016/s0306-4522(97)00157-7. Neuroscience. 1997. PMID: 9300402
-
Stimulation of the rat mesolimbic dopaminergic system produces a pressor response which is mediated by dopamine D-1 and D-2 receptor activation and the release of vasopressin.Brain Res. 1995 Dec 1;701(1-2):28-38. doi: 10.1016/0006-8993(95)00967-x. Brain Res. 1995. PMID: 8925292
-
Role of the mesolimbic dopamine system in cardiovascular homeostasis. Stimulation of the ventral tegmental area modulates the effect of vasopressin on blood pressure in conscious rats.Clin Exp Pharmacol Physiol. 1998 Sep;25(9):661-8. doi: 10.1111/j.1440-1681.1998.tb02273.x. Clin Exp Pharmacol Physiol. 1998. PMID: 9750953 Review.
-
Inhibition of cardiac baroreflex sensitivity after central dopaminergic stimulation.Clin Exp Pharmacol Physiol. 1998 Jul-Aug;25(7-8):624-6. doi: 10.1111/j.1440-1681.1998.tb02264.x. Clin Exp Pharmacol Physiol. 1998. PMID: 9673440
-
Behavioural effects of substance P through dopaminergic pathways in the brain.Ciba Found Symp. 1982;(91):307-24. doi: 10.1002/9780470720738.ch18. Ciba Found Symp. 1982. PMID: 6183078 Review.
Cited by
-
The ventral tegmental area as a putative target for tachykinins in cardiovascular regulation.Br J Pharmacol. 2005 Jul;145(6):712-27. doi: 10.1038/sj.bjp.0706249. Br J Pharmacol. 2005. PMID: 15895109 Free PMC article.
-
Blockade of tachykinin NK3 receptor reverses hypertension through a dopaminergic mechanism in the ventral tegmental area of spontaneously hypertensive rats.Br J Pharmacol. 2010 Dec;161(8):1868-84. doi: 10.1111/j.1476-5381.2010.01008.x. Br J Pharmacol. 2010. PMID: 20804497 Free PMC article.
-
Brain tissue integrity mapping in adults with obstructive sleep apnea using T1-weighted and T2-weighted images.Ther Adv Neurol Disord. 2022 Nov 19;15:17562864221137505. doi: 10.1177/17562864221137505. eCollection 2022. Ther Adv Neurol Disord. 2022. PMID: 36419869 Free PMC article.
-
Evaluation of GABA Receptors of Ventral Tegmental Area in Cardiovascular Responses in Rat.Iran J Med Sci. 2014 Jul;39(4):374-81. Iran J Med Sci. 2014. PMID: 25031490 Free PMC article.
-
Investigating the association between human brainstem microstructural integrity and hypertension using magnetic resonance relaxometry.Hypertens Res. 2025 Apr;48(4):1564-1574. doi: 10.1038/s41440-025-02114-1. Epub 2025 Jan 23. Hypertens Res. 2025. PMID: 39849049 Free PMC article.
References
-
- ABERCROMBIE E.D., KEEFE K.A., DIFRISCHIA D.S., ZIGMOND M.J. Differential effect of stress on in vivo dopamine release in striatum, nucleus accumbens, and medial prefrontal cortex. J. Neurochem. 1989;52:1655–1658. - PubMed
-
- BENNETT T., GARDINER S.M. Influence of exogenous vasopressin on baroreflex mechanisms. Clin. Sci. 1986;70:307–315. - PubMed
-
- BISHOP V.S., HAY M. Involvement of the area postrema in the regulation of sympathetic outflow to the cardiovascular system. Front Neuroendocrinol. 1993;14:57–75. - PubMed
-
- CECHETTO D.F., SAPER C.B. Role of the cerebral cortex in autonomic function Central regulation of autonomic functions 1990Oxford: Oxford University Press; 208–223.eds. Loewy, A.D. & Spyer, K.M.
-
- CORNISH J.L., VAN DEN BUUSE M. Pressor responses to electrical and chemical stimulation of the rat brain A10 dopaminergic system. Neurosci. Lett. 1994;176:142–146. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

